Sante Publique
Digestive poisoning , cholera is a notifiable disease caused by the ingestion of water or food contaminated by Vibrio cholerae bacilli of the toxigenic serogroups O1 and O139.
Cholera: the disease
A rare imported pathology in France
Cholera is an acute digestive poisoning caused by ingestion of water or food contaminated by Vibrio cholerae bacilli of toxigenic serogroups O1 and O139 (cholera vibrios).
Today, collective and individual sanitation and hygiene measures have led to the disappearance of cholera in France (excluding Guyana and Mayotte, where sporadic and limited epidemics were described in the decades 1990-2000 in the two previous decades). In mainland France, cholera, which is subject to mandatory notification , is indeed a rare imported pathology. Symptomatic cases of cholera are mainly linked to the absorption of contaminated drinks or food abroad.
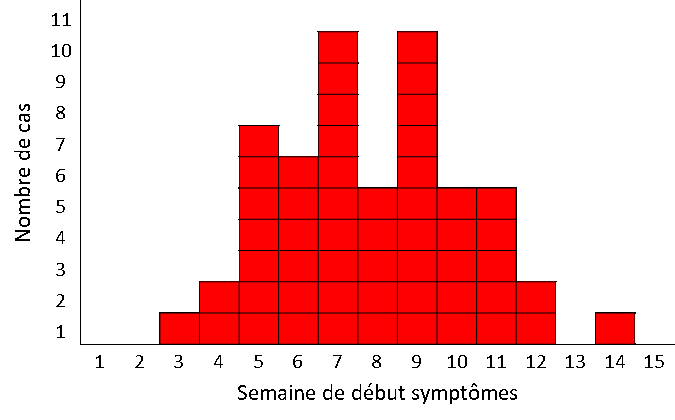
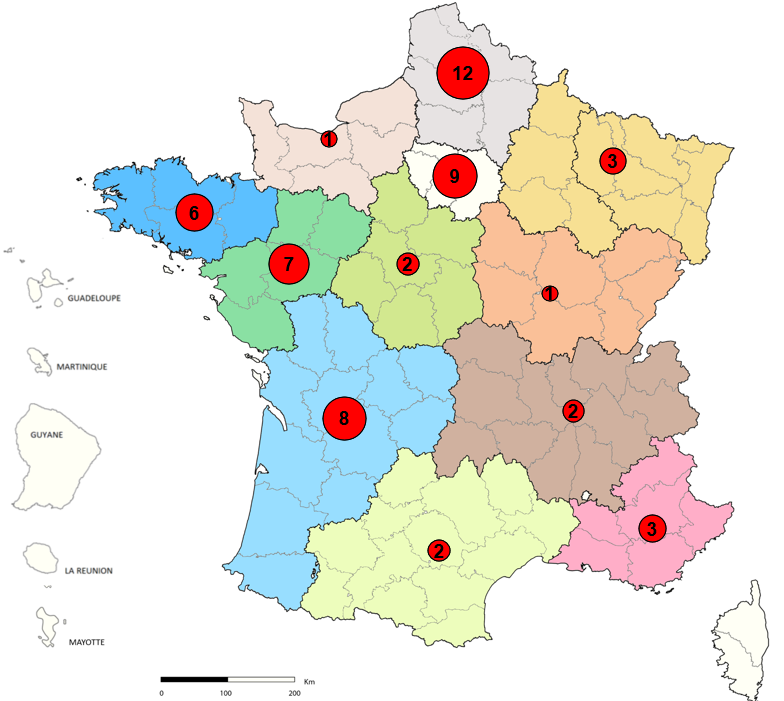
Early reporting of suspected and confirmed cases, and notification of confirmed cases takes place from a single case. They allow the management of imported cholera cases as soon as possible. Between 0 and 2 cases of cholera have been declared each year in France since 2000, they concern travelers returning from endemic areas . This is a small and decreasing number.
Although rare in France, cholera can cause severe digestive symptoms and dehydration. Even if the risks of contamination and epidemic are very limited on French territory, the epidemiological surveillance of Public Health France requires the early notification of cases as soon as this infection is suspected in order to prevent its spread.
Human-to-human and environmental transmission
Man is the main reservoir of cholera. But in some regions, the environment can also play this role, resulting in the circulation of cholera vibrios in an endemic mode . Cholera is linked to the absorption of contaminated water or food. Bacilli , or cholera vibrios, secrete cholera toxin in the intestine, which causes the loss of water and electrolytes (up to 15-20 liters per day). Diarrheal stools released in large quantities spread bacilli in the environment and faecal-oral transmission .
High population concentrations, combined with poor environmental hygiene, favor the appearance and development of cholera epidemics.
Prevention through hygiene and vaccination
When the basic rules of hygiene are respected, the vibrio responsible for cholera is not very transmissible. Appropriate chlorination of water and basic hygiene measures are generally sufficient to prevent contamination.
In the event of a trip to these endemic areas , compliance with hygiene measures (food hygiene with consumption of cooked and hot food, capsulated bottled water, avoidance of ice cubes, and hand washing) remains the best prevention.
There is no active vaccine against Vibrio cholerae serogroup O139. On the other hand, health personnel going to work with patients or in refugee camps during an epidemic can benefit from the anti-cholera vaccine (against different Vibrio cholerae O1 strains and a recombinant cholera toxin B subunit) administered orally (2 doses one week apart for adults and 3 doses one week apart for children 2 to 6 years of age).
Severe intestinal symptoms
The incubation period of cholera is short, from a few hours to five days.
Most people infected with Vibrio cholerae show few or no symptoms, although the bacillus can be found in their stool for one to two weeks. In case of illness, 80 to 90% of episodes are mild or moderately severe and it is then difficult to distinguish them clinically from other types of acute diarrhoea.
Less than 20% of patients develop all the typical symptoms of cholera, with symptoms of moderate to severe dehydration: violent diarrhea profuse with “rice water”, vomiting, without fever.
In the absence of treatment, death occurs in 1 to 3 days, by cardiovascular collapse in 25 to 50% of cases. Mortality is higher in children, the elderly and vulnerable individuals.
The diagnosis of cholera is clinical and biological. It is based on the detection of V. cholerae serogroup O1 or O139 producing cholera toxin in the stools of a patient. In the event of suspected isolation of a strain of cholera vibrio, immediate contact should be made with the National Reference Center for vibrios and cholera for typing and confirmation of the diagnosis: https://www.pasteur.fr/fr/ public-health/cnr/the-cnr/vibrions-cholera
A treatment based on rehydration
The treatment of cholera essentially consists of compensating for the digestive losses of water and electrolytes. Depending on the degree of dehydration, rehydration takes place orally or intravenously. An improvement in the subject’s condition is visible quickly (in a few hours) and healing occurs in a few days. There are no sequels. Antibiotic therapy can be useful in certain severe cases, but multi-resistant strains may appear.
An endemic circulation in South Asia
Cholera is regularly the subject of epidemics in developing countries where it evolves according to the country on an endemic and/or epidemic mode .
The world has been experiencing the seventh cholera pandemic since 1961, caused by Vibrio cholerae serogroup O1 .
Cholera has been endemic in the Indian subcontinent for several centuries. Cholera spread from 1817 to all of Asia, the Middle East and part of Africa, during the first cholera pandemic. Subsequent pandemics also developed from Asia and were facilitated by improved transportation. The seventh pandemic started in Indonesia in 1961, affected Asia in 1962, the Middle East and part of Europe in 1965, Africa in 1970, and South America in 1991.
On the n the island of Hispaniola an epidemic has been ongoing since the emergence of V. cholerae serogroup O1 in 2010 in Haiti.
Today, Africa and Asia are the two areas most affected by cholera. The disease is spreading there. The outbreak declared in Yemen since 2016 is the largest ever documented.
In 1992, a strain of Vibrio cholerae belonging to the new serogroup O139 appeared in India and Bangladesh. Since then, it has caused epidemics in several Asian countries and could one day be the cause of an eighth pandemic. Cholera is the first disease to have been the subject of international notification (since 1892).