FDA
As part of our ongoing efforts to combat foodborne illness and aligned with our New Era of Smarter Food Safety initiative, today, the U.S. Food and Drug Administration released the Cyclospora Prevention, Response and Research Action Plan. Modeled after our Leafy Greens Shiga toxin-producing E. coli (STEC) Action Plan, the plan focuses on improving prevention, enhancing response activities and filling knowledge gaps in order to help prevent Cyclospora contamination of foods and to help prepare for responding to future outbreaks.
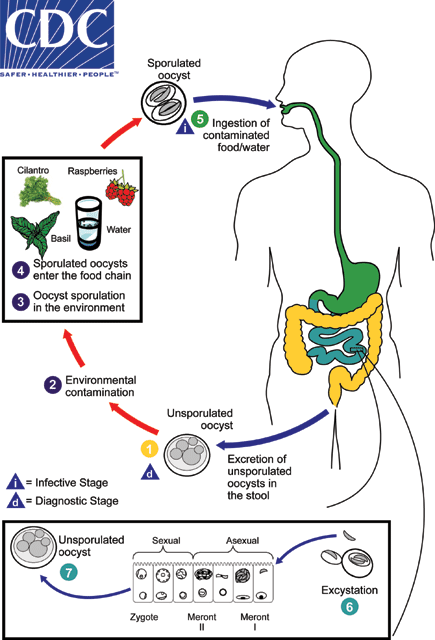
Cyclosporiasis is a foodborne intestinal illness caused by Cyclospora cayetanensis. The most common symptoms of cyclosporiasis are diarrhea, weight loss, nausea and fatigue. Cyclospora is historically associated with imported produce or travel outside the U.S.; however, we have also detected Cyclospora in domestically produced foods in recent years.
The FDA first documented Cyclospora in domestically grown produce (cilantro) in 2018 as part of an ongoing sampling assignment of fresh herbs. The testing was done as part of a surveillance sampling, and the cilantro sample was not linked to any illnesses or outbreaks. Not long after, domestically grown produce was again associated with an outbreak linked to a salad mix. During the FDA’s investigations into this outbreak, Cyclospora was also detected in an unused package of the salad containing domestically grown produce.
The availability of new testing methods for Cyclospora developed at the FDA’s Center for Food Safety and Applied Nutrition played an important role in helping the FDA identify these positive samples of Cyclospora in the cilantro and in the salad mix. The number of reported cases of this foodborne illness has been rising in recent years, in part because of better diagnostic and detection methods. According to the Centers for Disease Control and Prevention, there have been roughly 6,000 domestically acquired cases of Cyclospora over the last three years. The number of reported cases typically rises during the spring and summer, usually in May, June and July. Rising case numbers and the emergence of Cyclospora contamination in domestically grown produce prompted the FDA to create the Cyclospora Task Force in 2019. The task force is comprised of multidisciplinary experts across the FDA and CDC, with the goal of reducing the public health burden of foodborne illness caused by Cyclospora in produce.
The task force formulated the action plan announced today, which will serve as a strategic guide to improve prevention, enhance response activities and fill knowledge gaps about the presence of Cyclospora in or on foods. In the area of prevention, the new action plan highlights how we’re addressing this food safety issue through the development and delivery of prevention-focused education materials and outreach to stakeholders. We’re also working with industry to encourage the development of rapid test kits to specifically detect Cyclospora to better facilitate industry testing and root cause analysis activities. In addition, we plan to collaborate with industry to look for ways to more effectively control Cyclospora in the environment and on farms.
In the area of response, the plan is focused on expanding laboratory capacity across the FDA, state, foreign partners and academia to sample and test for Cyclospora, providing greater capacity to investigate during outbreak events. The FDA is also developing a new investigational tool to help guide assessments of farms potentially implicated in a Cyclospora outbreak to determine potential sources and routes of contamination.
Given the emerging nature of Cyclospora contamination in domestic produce, a large number of action items in this plan are aimed at addressing knowledge gaps. In this area, the FDA intends to work with CDC to better understand the case distribution of cyclosporiasis across the U.S. and to advance genotyping methods in clinical, food and environmental samples that will allow us to genetically link clinical cases to food products and environments identified by traceback during an outbreak. As part of this plan, the FDA also intends to work with industry and academia to better understand the prevalence of Cyclospora in agricultural water and to encourage data sharing to help stakeholders identify trends associated with Cyclospora contamination. Our action plan underscores the importance of collaboration across industry, academia, state, federal and foreign government partners, and other stakeholders to reduce the risks and public health burden caused by foodborne Cyclospora. We’ll continue updating this plan as actions are completed and new actions are identified.
While the U.S. has one of the safest food supplies in the world, we are focused on making it better. The FDA is focused on working with all stakeholders to achieve the goals identified in this Cyclospora Prevention, Response and Research action plan.