Archives
-
Join 346 other subscribers
KSWFoodWorld
Blog Stats
- 448,143 Views
Category Archives: Food Microbiology Research
Listeria monocytogenes in ready-to-eat (RTE) foods: attribution, characterization and monitoring
Executive summary
A virtual meeting of the Joint FAO/WHO Expert Meeting on Microbiological Risk Assessment (JEMRA) of Listeria monocytogenes (hereinafter referred to as “L. monocytogenes”) in ready-to-eat (RTE) foods: attribution, characterization and monitoring was held from 20 October to 6 November 2020.
The purpose of the meeting was to review recent data on L. monocytogenes and determine the need to modify, update, or develop new risk assessment models and tools for this pathogen. A public call for data and experts was issued to support this work. In addition, background documents on the various aspects related to the meeting were prepared ahead of time for consultation by the experts.
Prepared documents included the following:
1) assessment of past JEMRA documentation; “Risk assessment of Listeria monocytogenes in ready to eat foods: Interpretative summary (MRA4)” (FAO and WHO, 2004a) and “Risk assessment of Listeria monocytogenes in ready to eat foods: Technical report” (MRA5) (FAO and WHO, 2004b);
2) a review of current national L. monocytogenes surveillance programmes;
3) a review of current microbiological and laboratory methods for L. monocytogenes;
4) an update on the virulence markers for L. monocytogenes. The meeting participants reviewed the prepared summary documents and other information on outbreaks and disease attribution, virulence, population risk factors, advances in laboratory methods and surveillance.
The aforementioned risk assessment documents (MRA4, MRA5) (FAO and WHO, 2004a, 2004b) covered a cross-section of RTE foods (pasteurized milk, ice cream, cold smoked fish and fermented meats) linked to invasive listeriosis. Since the publication of these documents, outbreaks of listeriosis continue to occur across the globe associated with previously reported foods, but also with many previously unreported food vehicles, including fresh and minimally processed fruits and vegetables (e.g. frozen vegetables).
The expert group concluded that it would be wise to be more inclusive in future risk assessments and that a full farm-to-fork risk assessment be considered. L. monocytogenes can infect anyone; however, it continues to disproportionally affect certain highly susceptible populations. The expert group recommended that future risk assessments should review groupings of susceptible groups, based on physiological risks and other socio-economic factors.
New information has emerged on L. monocytogenes strain variants, which differ in their virulence and environmental tolerance. Based on a panel of specific genes, the expert group proposed a virulence ranking of L. monocytogenes relevant
to invasive listeriosis.
The expert group concluded that the development and implementation of effective surveillance systems are critical in addressing the control of L. monocytogenes. The use of approved standardized laboratory methods that culture and isolate strains should be the foundation so that human, food and environmental isolates can be further characterized and inventoried.
In conclusion, the expert group identified several critical gaps in the current FAO/WHO risk assessment model and collectively agreed that updating the model would be valuable for informing risk analysis strategies, including in low- and middle-income countries (LMICs). The experts prepared short examples from literature (Annex 1) to demonstrate and highlight several key principles that should be considered in the risk assessment for L. monocytogenes.
Posted in Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Listeria, Listeria monocytogenes, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Research
USA – Core Outbreak Table – Investigations of Foodborne Illness Outbreaks
What’s New
- A new outbreak of E. coli O157:H7 (reference #1121) in a not yet identified product has been added to the table and traceback has been initiated.
- For the outbreak of Salmonella Litchfield in seafood, (reference #1105), FDA issued an Outbreak Advisory on 10/19/2022.
- Based on CDC’s epidemiological investigation of two large multistate outbreaks of Cyclospora cayetanensis (reference #1080 and #1084), ill people reported eating a variety of leafy greens before becoming sick. For both investigations, CDC, FDA, and state and local partners conducted epidemiologic and traceback investigations and collected and analyzed product and environmental samples. All samples collected were reported as negative for Cyclospora. Due to the lack of additional detail in the epidemiological data and the absence of supporting evidence collected from traceback and sample collection, FDA could not identify a specific product as the source of either outbreak.
- For the outbreak of Salmonella Senftenberg (reference #1087) in a not identified product, one additional case was reported, the outbreak has ended, and FDA’s investigation has closed.
- For the adverse illness event series in frozen food (reference #1076), the outbreak has ended, and the FDA investigation has closed.
Posted in Cyclospora, Cyclosporiasis, E.coli O157, E.coli O157:H7, FDA, food bourne outbreak, Food Illness, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Poisoning, Foodborne Illness, foodborne outbreak, foodbourne outbreak, Illness, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, outbreak, Poisoning, Salmonella, STEC, STEC E.coli
UK – Listeriosis in England and Wales: summary for 2020
Main points for 2020
This report summarises the number, demographics and clinical outcomes of confirmed cases of listeriosis in England and Wales in 2020:
A total 124 cases of listeriosis were reported in England and Wales.
Incidence rates of listeriosis were highest in people aged 80 years and over.
Overall, the crude incidence of listeriosis was lower in men than women, but reported cases among men aged 60 to 69 were 4 times higher than in women aged 60 to 69.
Pregnancy associated infections accounted for a fifth of all reported cases and, a 34.8% of pregnancy-associated cases resulted in stillbirth or miscarriage.
Among non-pregnancy associated cases of listeriosis, death was reported for 29 cases (29.3%), of whom 17 (17.2%) were known to have listeriosis recorded as a cause of death on the death certificate.
Incidence of listeriosis varied geographically, with the lowest incidence in the North West (0.15 per 100,000 population) and the highest in the East Midlands (0.27 per 100,0000 population).
There were 2 listeriosis outbreaks investigated in England, including a national outbreak associated with smoked salmon.
Posted in food contamination, food death, Food Hazard, Food Hygiene, Food Inspections, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Pathogen, food recall, Food Safety, Food Safety Alert, Food Safety Management, Food Testing, Listeria, Listeria monocytogenes, listeriosis, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Research
Australia – Outbreak of gastro at Canberra doughnut shop believed to be caused by sick worker and poor hand hygiene practices – Norovirus
The investigation found the illness was caused by the spread of norovirus, a virus that often causes gastro and is spread through direct contact with an infected person or ingestion of faeces or vomit particles from an infected person.
It found no reports of gastro symptoms from customers in the store, making it unlikely a sick patron was the cause of the outbreak, and also noted food handlers on site did not provide stool samples for testing.
The report stated there was evidence collected on-site “suggestive of faecal contamination”, and the spread of the virus was most likely a result of a worker carrying the virus.
Posted in food bourne outbreak, Food Illness, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Virus, Foodborne Illness, foodborne outbreak, foodbourne outbreak, Illness, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Norovirus, outbreak, Virus
FDA Works to Protect Consumers from Foodborne Illness and other Adverse Events
A Conversation with Stic Harris, Conrad Choiniere, and Michael Rogers on Foodborne Illness and other Adverse Events
Each year, the FDA receives approximately 9,600 reports of foodborne illness or other adverse events related to FDA-regulated human food and dietary supplement products submitted by individual consumers, public health professionals, or industry.[1] In addition, the FDA’s Coordinated Outbreak Response & Evaluation (CORE) Network evaluates an average of 75 incidents annually that may potentially constitute a national, multi-state outbreak.
The FDA takes its responsibility to safeguard the food supply very seriously and is committed to being transparent about how FDA approaches its food safety mission, particularly about how the agency responds when it receives reports of foodborne illness and other adverse events.
That is what three foods program leaders will discuss in this conversation about challenges and opportunities they face every day in working to protect consumers from potential hazards in the complex, global food system.
Stic Harris, DVM, MPH, is the director of FDA’s Coordinated Outbreak Response & Evaluation (CORE) Network, which is on the frontline of foodborne illness outbreak investigations.
Conrad Choiniere, Ph.D., is the director of the Office of Analytics and Outreach at FDA’s Center for Food Safety and Applied Nutrition (CFSAN). His office evaluates consumer complaints and reports of potential hazards associated with FDA-regulated food, dietary supplements and cosmetic products.
Michael Rogers, MS, is the Assistant Commissioner for Human and Animal Food Operations in FDA’s Office of Regulatory Affairs (ORA), the inspection and enforcement arm of the FDA. In his role he works with ORA’s consumer complaint coordinators and investigators in the field, who are the agency’s boots on the ground in this work to keep foods safe.
These three leaders represent different aspects of the agency’s food safety work and, together, they will provide an overview of how FDA is doing its job and what it can, and will, do to continually improve in this space.
Research -Food Safety Issues Related to Eating In and Eating Out
Abstract
Because of growing urbanization and lack of time to prepare meals at home, eating out or getting food delivered have become common trends for many people. The consumption of food from unknown sources may impose an increased chance of contamination with microbiological hazards, especially if sanitary conditions are not met. We evaluated data from health surveillance agencies and scientific articles on foodborne diseases (FBD) reported internationally according to the exposure sites. We observed that the data are influenced by cultural, political, and socioeconomic differences. For instance, in New Zealand, Australia, United States, Denmark and India, the occurrence of FBD outbreaks was greater from foods prepared in commercial establishments and street vendors than from households. Conversely, in China, countries of the European Union and Brazil, the results are the opposite. Additionally, the pandemic imposed new eating behaviour patterns, increasing delivery services and foods prepared in so-called “Dark Kitchens”. The underreporting and heterogeneity of data among countries prevented a precise conclusion to the question of whether homemade foods are inherently safer than foods prepared out. Nevertheless, a lower level of development in a country influences its sanitation conditions, as well as the number of street food vendors, the search for cheaper foods, and insufficient knowledge of the population on good hygiene practices, which can all increase the chances of FBD cases.
Research – Illnesses Linked to Harmful Algal Blooms

Highlight
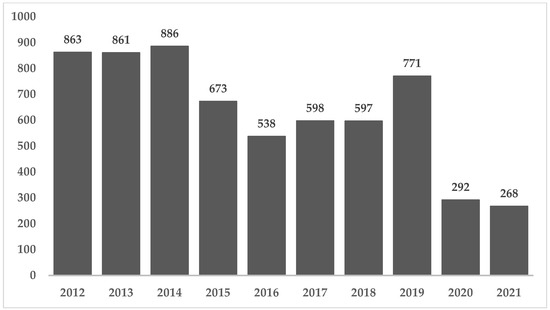
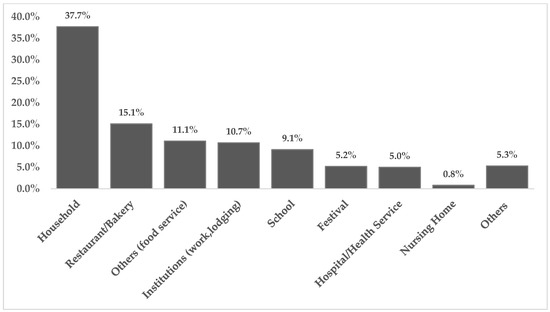
- Thirteen states reported 227 harmful algal blooms (HABs) that resulted in a total of 95 human illnesses and at least 1,170 animal illnesses.
- The first human death reported in OHHABS was associated with paralytic shellfish poisoning.
- A HAB event in September killed at least 1,000 fish (carp).
- 22 human illnesses (23%) were associated with national parks, with 21 illnesses attributed to a single HAB event.
Background
Harmful algal blooms (HABs) that result from the rapid growth of algae or cyanobacteria (sometimes referred to as blue-green algae) in natural waterbodies can harm people, animals, or the environment. HAB events of public health concern are primarily caused by microalgae called diatoms and dinoflagellates, cyanobacteria, and the toxins they can produce. HAB events, which can be intensified by factors such as nutrient pollution and warmer water temperature, can have public health, environmental, and economic impacts.
HABs are a One Health issue—they affect the health of people, animals, and our shared environment. One Health is a collaborative and multi-sectoral approach that involves engagement across disciplines including public health, animal health, and environmental health. Using a One Health approach, CDC collects data about HAB events and associated human or animal illnesses through the One Health Harmful Algal Bloom System (OHHABS) to inform public health prevention efforts.
Within the context of OHHABS, the term HAB event describes the identification of a bloom or the detection of HAB toxins in water or food (i.e., absent a visual bloom). Human illnesses are reported individually. Animal illnesses are reported as single cases of illness or in groups, such as flocks of birds. The reporting system can link HAB event data with human or animal illness data. OHHABS uses standard definitions [PDF – 3 pages] to classify HAB events as suspected or confirmed and human or animal illness as suspected, probable, or confirmed.
OHHABS is available for voluntary reporting by public health agencies and their designated environmental health or animal health partners in the United States, District of Columbia, Federated States of Micronesia, Guam, Marshall Islands, Northern Mariana Islands, Palau, Puerto Rico, and U.S. Virgin Islands. Public health agencies use standard forms to report HAB events, human cases of illness, and animal cases of illness to OHHABS. Public health agencies do not need to submit all three types of forms to participate.
Data collected for HAB events include general information (e.g., observation date), geographic information, water body characteristics (e.g., salinity), observational characteristics (e.g., water color, scum), and laboratory testing. Data collected for cases of illness include general demographic characteristics, exposure information, signs and symptoms, medical care, and health outcomes. OHHABS is a dynamic electronic reporting system; data within individual reports are subject to change over time. Data included in this report are from a specific point in time.
Posted in Algal Blooms, Algal Toxin, Food Illness, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Foodborne Illness, Illness, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk
New Zealand – Hepatitis A Outbreak linked to frozen berries jumps to 18.
The Ministry of Health said on Thursday afternoon that the number of confirmed cases linked to the outbreak had grown to 18 in total. Of these, nine (50%) have been hospitalized.
As of Thursday, 14 of the 18 cases have been sequenced to date – all 14 have an identical sequence profile, indicating they likely came from the same source.
Hepatitis A is spread by contact with feces of an infected person. It can be passed on through poor personal hygiene (such as when people don’t wash their hands properly), contaminated food and close personal contact.
All 18 cases reported “significant” consumption of uncooked, imported frozen berries.
Food safety officials have advised that Pams – a New Zealand division of supermarket giant Foodstuffs – was recalling various imported frozen berry products as a precaution, over a possible link to recent cases of the contagious virus in fruit from Serbia.
Posted in Food Illness, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Virus, Foodborne Illness, Hepatitis A, Illness, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Virus
Research – What to Know About the Risk of Cronobacter in Powdered Infant Formula
The U.S. Food and Drug Administration continues to help ensure the safety of powdered infant formula in the wake of reports of Cronobacter sakazakii infections in four infants that may have contributed to death in two patients.
Those reports led to an FDA investigation of Abbott Nutrition in Sturgis, Michigan. Insanitary conditions found at the facility led to the February 2022 voluntary recall of specific lots of powdered infant formula manufactured at the facility and brought attention to a type of bacteria that many parents may not have heard about before and scientists continue to work to better understand.
While Cronobacter contamination and infections are rare, it is still important to know the risks and what you can do to minimize them.
What Is Cronobacter?
Cronobacter is a germ (bacteria) that is naturally found in the environment. Cronobacter can exist on almost any surface and is especially good at surviving in dry foods, like powdered infant formula, powdered milk, herbal teas, and starches.
Cronobacter is harmless for most people and infections are rare. When they do occur, they can be life threatening in infants, particularly those who are younger than two months old, premature, immunocompromised, or of low birthweight.
How Does Powdered Infant Formula Become Contaminated with Cronobacter?
Parents may not know that while powdered infant formula manufacturers are required to produce formula that is safe from harmful bacteria like Cronobacter, powdered formulas — unlike liquid formulas — do not receive certain processing treatments that would render them sterile.
Cronobacter has the potential to come into factories, homes, and hospitals on the soles of shoes or on hands and can then live on surfaces like counters or bottles.
At the factory, Cronobacter could get into formula powder if:
- Contaminated ingredients are used to make the formula.
- The formula powder touches a contaminated surface.
In the home, infants can get infected by Cronobacter through multiple pathways including:
- Inadequately sanitized breast-pump accessories, bottles, and other utensils used for feeding.
- Inadequate handwashing prior to infant formula preparation.
- Formula lids or scoops placed on contaminated surfaces, like countertops or sinks, that then later touch the formula.
- Contaminated water used to mix formula.
Why Is it Difficult to Find the Source of Cronobacter Contamination?
Cronobacter sakazakii and its cousins in the broader Cronobacter species category are part of a large family of bacteria that are common in the environment — like in our yards, kitchens, and living rooms — so identifying the specific source of an illness or outbreak can be challenging.
Importantly, only one state in the U.S., Minnesota, currently requires that cases of Cronobacter sakazakii be reported to state public health authorities. As a result, many cases are unreported, and scientists have limited information about the genetic makeup of the bacteria found in cases that are reported. To provide some perspective, the National Center for Biotechnology Informatics (NCBI) database that tracks these pathogens has received roughly 480,000 submissions of genomic information for strains of Salmonella enterica compared to only 1086 submissions for strains of Cronobacter.
What Can You Do to Minimize the Risk of Cronobacter Contamination?
Safely Preparing and Storing Powdered Infant Formula
In most cases, it is safe to mix powdered infant formula following manufacturer’s instructions. If your baby is less than 2 months old, was born prematurely, or has a weakened immune system the Centers for Disease Control and Prevention (CDC) recommends using ready-to-feed infant formula. Liquid infant formula is made to be sterile (without germs) and is the safest option for infants not receiving breast milk. However, you can also take extra steps to prepare powdered formula for these infant groups by using hot water (at least 158°F/70°C) to help protect against Cronobacter and then cooling the formula before feeding your infant.
However, certain metabolic and specialty products include statements on their packaging warning consumers against heating because heating the particular product above 100°F could result in a loss of vitamins and nutrients. Therefore, caregivers should be especially mindful of the manufacturer’s instructions included on the packaging for specialty metabolic formulas. If the instructions prevent a consumer from heating their formula to at least 158°F, then caregivers should instead bring the water used to mix the formula to body temperature and follow the instructions in this printable infographic to prepare and store powdered infant formula safely.
Parents who suspect their infant became ill due to infant formula may call the FDA’s consumer hotline at 1-800-FDA-1088.
Other Tips to Minimize the Risk of Cronobacter Contamination
Infants could also be exposed to Cronobacter from unclean breast pumps or unclean bottles. To help keep your baby safe, you can help prevent Cronobacter from growing on these items. Keep your baby’s food, whether breast milk or formula, safe by carefully cleaning, sanitizing, and storing bottles and breast pump parts.
It is also important to wash your hands with soap and water, especially before preparing bottles and feeding. The CDC says that alcohol-based hand sanitizer with at least 60% alcohol can also be used.
What Are the Symptoms of Cronobacter infection?
Symptoms of infection in infants may start with fever, poor feeding, excessive crying, and/or very low energy. Some infants may also have seizures. Babies with these symptoms should be taken to a medical provider as soon as possible.
Posted in Cronobacter sakazakii, Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Safety, Food Safety Management, food safety training, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk
USA – Kerry’s former quality assurance director pleads guilty to Honey Smack outbreak – Salmonella
A fast-moving, but somewhat secretive federal prosecution has extracted guilty pleas for three misdemeanors from the quality assurance director for a food manufacturer working for the Kellogg Company.
The former quality assurance director pleaded guilty on Oct. 21 to three misdemeanor counts of Introducing adulterated food into Interstate Commerce. It was part of a plea deal that is among a half dozen documents in the case that are sealed, and therefore not available for public viewing. The federal Magistrate for the Central District of Illinois, Jonathan P. Hawley, has scheduled sentencing for Jan. 30. 2023.
The charges stem from the multistate outbreak of Salmonella Mbandaka infections from Kellogg’s Honey Smacks cereal, which sickened 135 people in 35 states in 2018. The infections required hospitalizations for 34 patients. The Kellogg Company recalled Honey Smacks cereal on June 14, 2018.
Posted in Decontamination Microbial, food bourne outbreak, Food Illness, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Foodborne Illness, foodborne outbreak, foodbourne outbreak, Illness, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, outbreak, Salmonella


