Brand : Similac
Name : Elecare
Reason for reporting : Recall due to microbiological risk
Publication date : 23 February 2022
Click to access C_17_PubblicazioneRichiami_1657_azione_itemAzione0_files_itemFiles0_fileAzione.pdf
Brand : Similac
Name : Elecare
Reason for reporting : Recall due to microbiological risk
Publication date : 23 February 2022
Click to access C_17_PubblicazioneRichiami_1657_azione_itemAzione0_files_itemFiles0_fileAzione.pdf
Posted in Cronobacter sakazakii, cross contamination, food contamination, food handler, Food Hazard, Food Hygiene, Food Inspections, Food Micro Blog, Food Microbiology, Food Microbiology Research, Food Microbiology Testing, Food Pathogen, food recall, Food Safety, Food Safety Alert, Food Testing, Salmonella
Abbott has taken the precautionary step of recalling various batches of Elecare Similac and Alimentum Similac infant formula powders because of the possible presence of Salmonella and Cronobacter sakazakii. Both products are foods used for special medical purposes for infants, ordinarily to be used under medical supervision. This PRIN is an update to include the possible presence of an additional pathogen and amended date and batch codes.
| Elecare Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 28130Z20 |
| Best before | October 2022 |
| Elecare Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 29266Z20 |
| Best before | December 2022 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 27937Z26 |
| Best before | March 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 28123Z26 |
| Best before | April 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 29210Z20 |
| Best before | May 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 29280Z26 |
| Best before | May 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 30283Z26 |
| Best before | June 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 30281Z26 |
| Best before | June 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 31410Z20 |
| Best before | July 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 31411Z20 |
| Best before | July 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 33688Z20 |
| Best before | September 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 32431Z20 |
| Best before | October 2023 |
| Elecare Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 30374Z20 |
| Best before | December 2022 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 34929Z20 |
| Best before | October 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 35030Z20 |
| Best before | November 2023 |
| Elecare Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 32422Z21 |
| Best before | February 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 22333Z26 |
| Best before | October 2022 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 23443Z26 |
| Best before | November 2022 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 25550Z26 |
| Best before | January 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 25564Z26 |
| Best before | January 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 26867Z26 |
| Best before | February 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 27936Z26 |
| Best before | March 2023 |
The potential presence of Salmonella and Cronobacter sakazakii in the products listed above.
Symptoms caused by Salmonella and Cronobacter sakazakii usually include fever, diarrhoea and abdominal cramps, although in severe cases may lead to sepsis or meningitis which include symptoms in infants including poor feeding, irritability, temperature changes, jaundice (yellow skin and whites of the eyes) and abnormal breaths and movements.
Posted in Cronobacter sakazakii, cross contamination, food contamination, food handler, Food Hazard, Food Hygiene, Food Illness, Food Inspections, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Pathogen, food recall, Food Safety, Food Safety Alert, Food Testing, fsa, Salmonella
Meju, a raw material for doenjang preparation, is highly vulnerable to aflatoxin-producing fungi. The aim of this study was to evaluate the effect of a one-year fermentation on aflatoxins and aflatoxin-producing fungi in doenjang spiked with aflatoxins B1, G1, B2, and G2 and inoculated with toxigenic Aspergillus flavus. A significant reduction in aflatoxins was observed after a year of fermentation, measuring 92.58%, 100%, 98.69%, and 100% of B1, G1, B2, and G2, respectively. After a year of fermentation, 6.95 ± 3.64 µg/kg of total aflatoxin was detected, which represents a 97.88% reduction in the total aflatoxin compared with the initial value (328.83 ± 36.60 µg/kg). Several aflatoxin-degrading fungi (Aspergillus versicolor, Cladosporium subcinereum, Aspergillus ochraceus) and bacteria (Bacillus albus, Bacillus velezensis) isolated from doenjang were identified as the major contributors to the reduction of aflatoxin. Furthermore, it was observed that most of the aflatoxin contamination in doenjang occurred during the meju stage, and this stage was found to be most susceptible to A. flavus contamination and growth. These findings reveal that native microorganisms mediate aflatoxin clean-up in doenjang during fermentation and support the use of such microorganisms as a starter culture for the preparation of aflatoxin-free doenjang.
Posted in Aflatoxin, Aflatoxin B1, Aspergillus, Bacillus, Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, microbial contamination, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, mold, Mold Toxin, Mould Toxin, Mould/Mold, Moulds, Research
Food processing environment flooring can become contaminated with pathogens in many ways including foot and equipment traffic, incoming materials, and floor drain backups. Natural antimicrobial turmeric and commercially available powdered floor treatments may reduce the levels of pathogens on flooring thereby reducing the risk of cross contamination from the floor to food contact surfaces. These chemicals were evaluated to determine their effectiveness against cocktails of Salmonella , Escherichia coli , and Listeria monocytogenes dried onto the surfaces of carriers made from polyurethane-concrete commercial flooring material. Aqueous test solutions were prepared from the minimum treatment required per m 2 from the manufacturer’s instructions diluted in sterile water. Potential synergy between turmeric and a percarbonate based commercial floor treatment was explored with a mixture of turmeric and sodium percarbonate, each at approximately 37g/m 2 application rate. Each inoculated carrier was exposed to the treatment solutions or a sterile water control for 10 minutes at room temperature, neutralized with Hi-Cap neutralizing broth, the bacteria suspended, enumerated, and log 10 reductions calculated for each treatment and inoculum combination. Mean log 10 CFU/carrier reductions with standard deviations ranged between 4.29±0.34 for the sodium percarbonate (SPC) based treatment and 0.004±0.23 for turmeric for Salmonella , 4.81±0.16 for SPC based treatment and -0.16±0.62 for turmeric for E. coli , and 4.88±0.6 for SPC based treatment and -0.16±0.15 for turmeric for L. monocytogenes .
Posted in Decontamination Microbial, E.coli, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Technology, Listeria, Listeria monocytogenes, microbial contamination, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Research, Salmonella
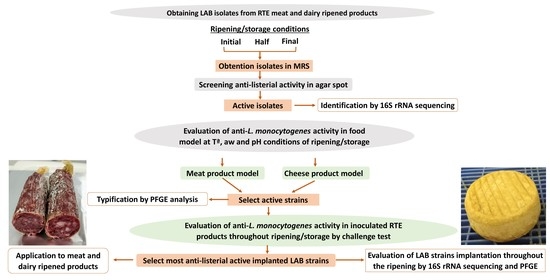
Listeria monocytogenes is one of the most important foodborne pathogens. This microorganism is a serious concern in the ready-to-eat (RTE) meat and dairy-ripened products industries. The use of lactic acid bacteria (LAB)-producing anti-L. monocytogenes peptides (bacteriocins) and/or lactic acid and/or other antimicrobial system could be a promising tool to control this pathogen in RTE meat and dairy products. This review provides an up to date about the strategies of use of LAB and their metabolites in RTE meat products and dairy foods by selecting the most appropriate strains, by analysing the mechanism by which they inhibit L. monocytogenes and methods of effective application of LAB, and their metabolites in these kinds of products to control this pathogen throughout the processing and storage. The selection of LAB with anti-L. monocytogenes activity allows to dispose of effective strains in meat and dairy-ripened products, achieving reductions form 2–5 logarithmic cycles of this pathogen throughout the ripening process. The combination of selected LAB strains with antimicrobial compounds, such as acid/sodium lactate and other strategies, as the active packaging could be the next future innovation for eliminating risk of L. monocytogenes in meat and dairy-ripened products.
Posted in Antibacterial, antimicrobial resistance, Antimicrobials, Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, LAB, lactic acid bacteria, Listeria, Listeria monocytogenes, microbial contamination, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Research
More than 30 people have reported becoming ill after they consumed food and/or drink at a northwest Las Vegas restaurant, according to the Southern Nevada Health District.
Secret of Siam, 5705 Centennial Center, has been closed as the health district investigates.
“The Health District has received reports from more than 30 people whose symptoms included increased heart rate, blurry vision, hallucinations, disorientation or confusion, dizziness or vertigo, loss of consciousness, dry mouth, and numbness and tingling in extremities within hours of consuming food or drink from Secret of Siam,” a health district news release stated.
The incidents occurred in January and February.
Anyone who may have eaten food at the restaurant is asked to take a survey at www.snhd.info/siamsurvey.
Posted in food bourne outbreak, Food Illness, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Foodborne Illness, foodborne outbreak, foodbourne outbreak, Illness, microbial contamination, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, outbreak
Studies on trimethylamine (TMA) in egg yolk have focused on how it impacts the flavor of eggs, but there has been little focus on its other functions. We designed an in vitro antibacterial test of TMA according to TMA concentrations that covered the TMA contents typically found in egg yolk. The change in TMA content in yolk was analyzed at different storage temperatures and for different storage durations. The known antibacterial components of eggs, including the cuticle quality of the eggshell and the lysozyme activity and content in egg white, were also assessed. The total bacterial count (TBC) of different parts of eggs were detected. The results showed that the inhibitory effect of TMA on Escherichia coli (E. coli) growth increased with increasing TMA concentration, and the yolk TMA content significantly increased with storage duration (p < 0.05). The cuticle quality and lysozyme content and activity significantly decreased with storage time and increasing temperature, accompanied by a significant increase in the TBC on the eggshell surface and in the egg white (p < 0.05). This work reveals a new role for trace TMA in yolks because it reduces the risk of bacterial colonization, especially when the antibacterial function of eggs is gradually weakened during storage.
Posted in Antibacterial, Antimicrobials, Decontamination Microbial, E.coli, escherichia coli, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Technology, microbial contamination, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Technology
To survive in hostile environments, bacteria attach to one another, forming a supportive framework known as a biofilm. In biofilms of Salmonella — a major cause of food-borne diarrheal illness — a key component of this framework is curli amyloid protein.
Now, in new research, scientists at the Lewis Katz School of Medicine at Temple University show that the repression of curli by an environmental factor in the intestine plays a critical role in freeing Salmonella bacteria of strain S. Typhimurium from their biofilms, enabling them to cause active infection. The environmental cue is nitrate, which both represses curli and modulates levels of an intracellular molecule known as cyclic-di-GMP. These events ultimately lead to the activation of S. Typhimurium flagella, which in humans is a critical step in allowing individual S. Typhimurium bacteria to swim toward and infect intestinal cells.
“It had been unclear what factors trigger S. Typhimurium to switch between a sessile, biofilm lifestyle to a motile, free-swimming lifestyle in the intestine,” explained Çagla Tükel, PhD, Director of the Center for Microbiology and Immunology at the Katz School of Medicine and senior investigator on the new study. “Our study shows for the first time that nitrate produced in the intestinal lumen of the host serves as an environmental cue driving this switch.”
Bovine tuberculosis, caused by Mycobacterium bovis (M. bovis), is a globally distributed chronic disease of animals. The bacteria can be transmitted to humans via the consumption of unpasteurised (raw) milk, thus representing an important public health risk. To investigate the risk of zoonotic transmission of M. bovis via raw milk, this study systematically reviewed published studies to estimate the prevalence of M. bovis in on-farm bulk-tank milk (BTM) and individual cow’s milk (IM) by meta-analysis.
In total, 1,339 articles were identified through seven electronic databases and initially screened using titles and abstracts. The quality of 108 potentially relevant articles was assessed using full texts, and 67 articles comprising 83 studies (76 IM and 7 BTM), were included in the meta-analysis. The prevalence of M. bovis in IM and BTM was summarised according to the diagnostic test used, and the tuberculin skin test (TST) infection status of the individual cows (for IM) or herds (for BTM). Heterogeneity was quantified using the I-squared statistic. Prediction intervals (95% PIs) were also estimated.
For IM, the overall prevalence was summarised at 5% (95%CI: 3%–7%). In TST positive cows, prevalence was summarised at 8% (95%CI: 4%–13%). For BTM, the overall prevalence independent of individual herd TST infection status was summarised at 5% (95%CI: 0%–21%).
There was considerable heterogeneity evident among the included studies, while PIs were also wide. Inconsistency in the quality of reporting was also observed resulting in missing information, such as the TST infection status of the individual animal/herd. No study reported the number of M. bovis bacteria in test-positive milk samples. Several studies reported the detection of M. tuberculosis and M. africanum in milk.
Despite international efforts to control tuberculosis, this study highlights the risk of zoonotic transmission of M. bovis via unpasteurised milk and dairy products made using raw milk.
Strawberries are an important fruit in Belgium both in production and consumption, but little information is available about the presence of Salmonella and STEC in these berries, the risk factors in agricultural production and possible specific mitigation options. In 2012, a survey was undertaken of three soil and three soilless cultivation systems in Belgium. No Salmonella spp. was isolated. No STEC was detected in the strawberry samples (0 out of 72), but STEC was detected by qPCR in 11 out of 78 irrigation water and 2 out of 24 substrate samples.
Culture isolates were obtained for 2 out of 11 qPCR positive irrigation water samples and 2 out of 2 substrate samples. Multivariable logistic regression analysis revealed elevated generic E. coli numbers (odds ratio (OR) for 1 log increase being 4.6) as the most important risk factor for STEC, together with the berry picking season (elevated risk in summer).
Presence of generic E. coli in the irrigation water (≥ 1 cfu per 100 ml) was mainly influenced by the type of irrigation water (collected rainfall water stored in ponds was more often contaminated than ground water pumped from boreholes (OR = 5.8)) and the lack of prior treatment (untreated water versus water subjected to sand filtration prior to use (OR = 19.2)).
The follow-up study in 2013 at one of the producers indicated cattle as the most likely source of STEC contamination of the irrigation water.
Posted in Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Technology, microbial contamination, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Research, Salmonella, STEC, STEC E.coli, Technology