Brand : Similac
Name : Elecare
Reason for reporting : Recall due to microbiological risk
Publication date : 23 February 2022
Click to access C_17_PubblicazioneRichiami_1657_azione_itemAzione0_files_itemFiles0_fileAzione.pdf
Brand : Similac
Name : Elecare
Reason for reporting : Recall due to microbiological risk
Publication date : 23 February 2022
Click to access C_17_PubblicazioneRichiami_1657_azione_itemAzione0_files_itemFiles0_fileAzione.pdf
Posted in Cronobacter sakazakii, cross contamination, food contamination, food handler, Food Hazard, Food Hygiene, Food Inspections, Food Micro Blog, Food Microbiology, Food Microbiology Research, Food Microbiology Testing, Food Pathogen, food recall, Food Safety, Food Safety Alert, Food Testing, Salmonella
Do not use, sell, serve or distribute the affected product.
Certain Raw BC Oysters recalled due to norovirus.
The recalled product has been sold in British Columbia.
| None | Raw BC Oysters | Variable – Sold by the dozen | LF 1402294 Subarea 14-8 Harvested January 31, 2022 Lot# 22020135 |
Waitrose is recalling Waitrose & Partners Green Pesto with Basil because salmonella has been found in the product.
| Waitrose & Partners Green Pesto with Basil | |
|---|---|
| Pack size | 145g |
| Use by | 27 February 2022 |
The presence of salmonella in the product listed above.
Symptoms caused by salmonella typically include fever, diarrhoea, and abdominal cramps.
Abbott has taken the precautionary step of recalling various batches of Elecare Similac and Alimentum Similac infant formula powders because of the possible presence of Salmonella and Cronobacter sakazakii. Both products are foods used for special medical purposes for infants, ordinarily to be used under medical supervision. This PRIN is an update to include the possible presence of an additional pathogen and amended date and batch codes.
| Elecare Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 28130Z20 |
| Best before | October 2022 |
| Elecare Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 29266Z20 |
| Best before | December 2022 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 27937Z26 |
| Best before | March 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 28123Z26 |
| Best before | April 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 29210Z20 |
| Best before | May 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 29280Z26 |
| Best before | May 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 30283Z26 |
| Best before | June 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 30281Z26 |
| Best before | June 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 31410Z20 |
| Best before | July 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 31411Z20 |
| Best before | July 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 33688Z20 |
| Best before | September 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 32431Z20 |
| Best before | October 2023 |
| Elecare Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 30374Z20 |
| Best before | December 2022 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 34929Z20 |
| Best before | October 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 35030Z20 |
| Best before | November 2023 |
| Elecare Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 32422Z21 |
| Best before | February 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 22333Z26 |
| Best before | October 2022 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 23443Z26 |
| Best before | November 2022 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 25550Z26 |
| Best before | January 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 25564Z26 |
| Best before | January 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 26867Z26 |
| Best before | February 2023 |
| Alimentum Similac | |
|---|---|
| Pack size | 400g |
| Batch code | 27936Z26 |
| Best before | March 2023 |
The potential presence of Salmonella and Cronobacter sakazakii in the products listed above.
Symptoms caused by Salmonella and Cronobacter sakazakii usually include fever, diarrhoea and abdominal cramps, although in severe cases may lead to sepsis or meningitis which include symptoms in infants including poor feeding, irritability, temperature changes, jaundice (yellow skin and whites of the eyes) and abnormal breaths and movements.
Posted in Cronobacter sakazakii, cross contamination, food contamination, food handler, Food Hazard, Food Hygiene, Food Illness, Food Inspections, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Pathogen, food recall, Food Safety, Food Safety Alert, Food Testing, fsa, Salmonella
Brand: Similac
Name: Alimentum
Reason for reporting: Recall due to microbiological risk Publication date: 23 February 2
Click to access C_17_PubblicazioneRichiami_1658_azione_itemAzione0_files_itemFiles0_fileAzione.pdf
Posted in Cronobacter sakazakii, cross contamination, food contamination, food handler, Food Hazard, Food Hygiene, Food Inspections, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Testing, Food Pathogen, food recall, Food Safety, Food Safety Alert, Food Testing, Salmonella
Tesco is recalling Tesco Hog Roast Style Pork with Apple Sauce because the crackling crumb sachet within the pack may contain Salmonella.
| Tesco Hog Roast Style Pork with Apple Sauce (suitable for freezing) | |
|---|---|
| Pack size | 425g |
| Use by | 06 January 2022, 09 January 2022, 15 January 2022, 17 January 2022, 6 February 2022, 8 March 2022, 9 March 2022 |
The possible presence of salmonella in the product listed above.
Symptoms caused by salmonella usually include fever, diarrhoea and abdominal cramps
The Food Standards Agency (FSA) has welcomed the ban after a link was confirmed between a Salmonella outbreak in people and feeder rodents used for reptile food originating from a premises in Lithuania.
The ban, imposed until further notice by the Department for Environment, Food and Rural Affairs comes after a joint investigation by UKHSA, FSA, DEFRA and APHA into an outbreak of salmonella affecting over 900 people in the UK. The FSA and partners are continuing to urge people to be extra careful when handling any frozen rodents including mice product and packaging due to the risk of salmonella.
People should be extra vigilant, washing hands thoroughly with soap and water immediately after contact, when handling not just the product, but their reptiles and associated equipment and environment, due to the risk of the illness.
Further general advice on reducing the risk of contracting Salmonella (Opens in a new window) (Opens in a new window)is available online.
Tina Potter, Head of Incidents at the Food Standards Agency said:
“As we have continued to see a rise in the number of cases of Salmonella Enteritidis linked to feeder rodents imported from Lithuania over the past number of months, we welcome Defra’s move to ban these products from being imported and sold across the UK.
Even though this ban has been introduced to ensure public health is protected, we cannot emphasise enough the importance of good hygiene practice when handling raw or frozen pet food, as well as the reptile itself”
The feed should be suitably stored, ideally in a dedicated storage compartment or freezer, not in contact with human food and it should always be defrosted naturally at room temperature on newspaper or paper towels away from human food and food preparation surfaces. Any surfaces and equipment used should be thoroughly disinfected.
Handlers and pet owners must always wash their hands thoroughly with soap and warm water immediately after handling the frozen and defrosted feed and handling your reptile and their equipment.”
Advice to reptile owners about feeding their pets
Snake owners and others using frozen mice as food may have concerns about maintaining their animal’s welfare, as the import ban may cause short term shortages. There should be sufficient mice to maintain animal welfare for all snakes and other animals, including birds that need to be fed mice, if owners adapt their current feeding routines. Detailed advice for reptile owners (Opens in a new window) (Opens in a new window)will be published online.
Advice to parents and guardians of children handling reptiles
Children have been particularly affected so we are urging parents and guardians to make sure everyone washes their hands thoroughly with warm soapy water every time they handle and feed mice to their pets and handle their reptiles to reduce risk of becoming ill with Salmonella. Both the vivarium and the areas reptiles are able to roam could be contaminated with Salmonella. Good hygiene should be observed.
If you, or other family members become ill with symptoms such as diarrhoea, abdominal pain and fever, consult your doctor or NHS 111 and inform them that you own/keep a reptile. If you have symptoms, make sure you wash your hands regularly and avoid preparing food for others. Do not go to work or school until 48 hours after symptoms have passed to reduce the chances of passing on the infection.
Meju, a raw material for doenjang preparation, is highly vulnerable to aflatoxin-producing fungi. The aim of this study was to evaluate the effect of a one-year fermentation on aflatoxins and aflatoxin-producing fungi in doenjang spiked with aflatoxins B1, G1, B2, and G2 and inoculated with toxigenic Aspergillus flavus. A significant reduction in aflatoxins was observed after a year of fermentation, measuring 92.58%, 100%, 98.69%, and 100% of B1, G1, B2, and G2, respectively. After a year of fermentation, 6.95 ± 3.64 µg/kg of total aflatoxin was detected, which represents a 97.88% reduction in the total aflatoxin compared with the initial value (328.83 ± 36.60 µg/kg). Several aflatoxin-degrading fungi (Aspergillus versicolor, Cladosporium subcinereum, Aspergillus ochraceus) and bacteria (Bacillus albus, Bacillus velezensis) isolated from doenjang were identified as the major contributors to the reduction of aflatoxin. Furthermore, it was observed that most of the aflatoxin contamination in doenjang occurred during the meju stage, and this stage was found to be most susceptible to A. flavus contamination and growth. These findings reveal that native microorganisms mediate aflatoxin clean-up in doenjang during fermentation and support the use of such microorganisms as a starter culture for the preparation of aflatoxin-free doenjang.
Posted in Aflatoxin, Aflatoxin B1, Aspergillus, Bacillus, Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, microbial contamination, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, mold, Mold Toxin, Mould Toxin, Mould/Mold, Moulds, Research
Food processing environment flooring can become contaminated with pathogens in many ways including foot and equipment traffic, incoming materials, and floor drain backups. Natural antimicrobial turmeric and commercially available powdered floor treatments may reduce the levels of pathogens on flooring thereby reducing the risk of cross contamination from the floor to food contact surfaces. These chemicals were evaluated to determine their effectiveness against cocktails of Salmonella , Escherichia coli , and Listeria monocytogenes dried onto the surfaces of carriers made from polyurethane-concrete commercial flooring material. Aqueous test solutions were prepared from the minimum treatment required per m 2 from the manufacturer’s instructions diluted in sterile water. Potential synergy between turmeric and a percarbonate based commercial floor treatment was explored with a mixture of turmeric and sodium percarbonate, each at approximately 37g/m 2 application rate. Each inoculated carrier was exposed to the treatment solutions or a sterile water control for 10 minutes at room temperature, neutralized with Hi-Cap neutralizing broth, the bacteria suspended, enumerated, and log 10 reductions calculated for each treatment and inoculum combination. Mean log 10 CFU/carrier reductions with standard deviations ranged between 4.29±0.34 for the sodium percarbonate (SPC) based treatment and 0.004±0.23 for turmeric for Salmonella , 4.81±0.16 for SPC based treatment and -0.16±0.62 for turmeric for E. coli , and 4.88±0.6 for SPC based treatment and -0.16±0.15 for turmeric for L. monocytogenes .
Posted in Decontamination Microbial, E.coli, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Technology, Listeria, Listeria monocytogenes, microbial contamination, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Research, Salmonella
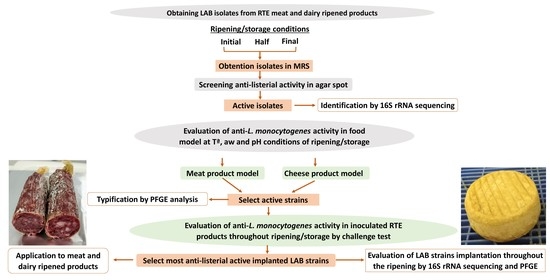
Listeria monocytogenes is one of the most important foodborne pathogens. This microorganism is a serious concern in the ready-to-eat (RTE) meat and dairy-ripened products industries. The use of lactic acid bacteria (LAB)-producing anti-L. monocytogenes peptides (bacteriocins) and/or lactic acid and/or other antimicrobial system could be a promising tool to control this pathogen in RTE meat and dairy products. This review provides an up to date about the strategies of use of LAB and their metabolites in RTE meat products and dairy foods by selecting the most appropriate strains, by analysing the mechanism by which they inhibit L. monocytogenes and methods of effective application of LAB, and their metabolites in these kinds of products to control this pathogen throughout the processing and storage. The selection of LAB with anti-L. monocytogenes activity allows to dispose of effective strains in meat and dairy-ripened products, achieving reductions form 2–5 logarithmic cycles of this pathogen throughout the ripening process. The combination of selected LAB strains with antimicrobial compounds, such as acid/sodium lactate and other strategies, as the active packaging could be the next future innovation for eliminating risk of L. monocytogenes in meat and dairy-ripened products.
Posted in Antibacterial, antimicrobial resistance, Antimicrobials, Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, LAB, lactic acid bacteria, Listeria, Listeria monocytogenes, microbial contamination, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Research