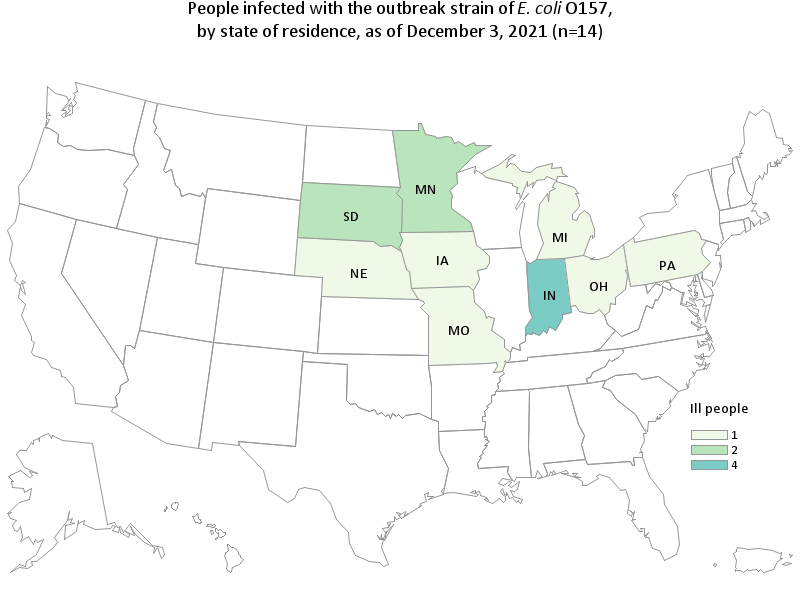
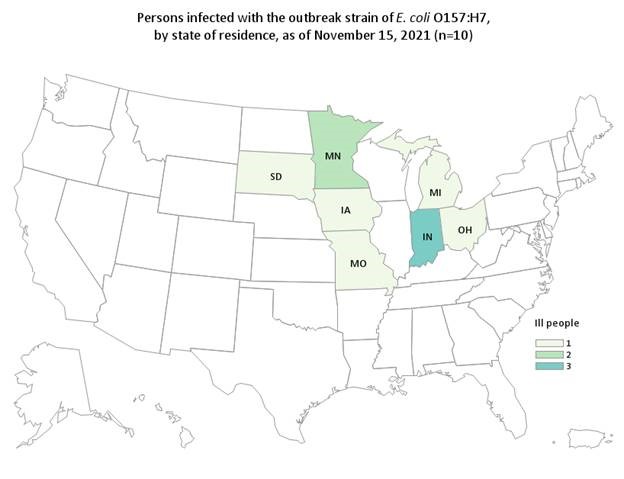
The FDA and CDC, in collaboration with state and local partners, are investigating illnesses in a multistate outbreak of E. coli O157:H7. According to the CDC, as of November 15, 2021, 10 people infected with the outbreak strain of E. coli O157:H7 have been reported from 7 states. Illnesses started on dates ranging from October 15 to October 27, 2021. Five people in this outbreak report eating spinach in the week before becoming sick and one person reported Josie’s Organics brand.
On November 15, 2021, the Minnesota Department of Agriculture reported that, as part of this outbreak investigation, a sample of Josie’s Organics Baby Spinach collected from the home of an ill person tested positive for E. coli O157:H7. This sample had a “Best If Used By” date of October 23, 2021 and is undergoing Whole Genome Sequencing (WGS) analysis to determine if the strain of E. coli O157:H7 present in the product sample matches the outbreak strain.
As WGS analysis of the sample is underway, FDA is tracing back the supply of the baby spinach in the positive product sample. Thus far, FDA has traced supply chains for this product back to a small number of farms in two different geographic regions and is deploying investigators along the supply chains of interest.
FDA and state partners are working with the firm to determine if additional products could be affected. This is an ongoing investigation and additional information will be provided as it becomes available.
Recommendation
Consumers, restaurants, and retailers, should not eat, sell, or serve Josie’s Organics Baby Spinach with a “Best If Used By” date of October 23, 2021. Josie’s Organics Baby Spinach is sold in a clear plastic clamshell with the Best If Used By Date on the top label.
Although this product is past expiration and should no longer be available for purchase, consumers should check their homes for product and discard it. If consumers froze fresh Josie’s Organics Baby Spinach, they should discard it.