Abstract

Posted in Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Listeria, Listeria monocytogenes, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Research
Posted in Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Listeria, Listeria monocytogenes, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Research
Posted in Bacteria, bacterial contamination, Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Pathogen, Food Technology, Listeria, Listeria monocytogenes, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Risk, Microwave Plasma Treatment, Pathogen, pathogenic, Research, Salmonella, Technology
Posted in Aflatoxin, Aflatoxin B1, Animal Feed Mould Toxin, Aspergillus Toxin, Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Toxin, Fusarium Toxin, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Mold Toxin, Mould Toxin, Mycotoxin, Ochratoxin, Ochratoxin A, Pet Food Aflatoxin, Research, Toxin
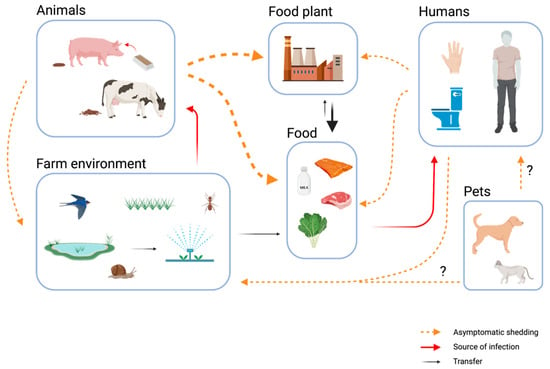
In winter, an increase in norovirus cases can be expected, as the infection is called the winter plague in some languages (eg vinterkräksjuka in Swedish). The main symptoms of norovirus infection are malaise, vomiting, diarrhea, fever and abdominal pain.
The virus is highly contagious and only a few viruses are needed to cause infection. Noroviruses can be transmitted through food from people who are ill or have recently been ill with norovirus. There are examples of norovirus being transmitted through food in Iceland and abroad, such as frozen raspberries, oysters and food from restaurants. Such infections can cause group infections.
For example, the consumption of frozen raspberries caused a widespread norovirus infection in Denmark a few years ago. Subsequently, the Danes established rules that frozen raspberries should be heated before consumption, and such instructions can often be seen on packaging.
But how can the virus be prevented from spreading to food at home, in canteens, restaurants and other food businesses?
The risk of norovirus being transmitted through food can be reduced if these guidelines are followed.
Posted in Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Virus, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Norovirus, Research, Virus
Interpretation of whole-genome sequencing (WGS) data for foodborne outbreak investigations is complex, as the genetic diversity within processing plants and transmission events need to be considered. In this study, we analyzed 92 food-associated Listeria monocytogenes isolates by WGS-based methods. We aimed to examine the genetic diversity within meat and fish production chains and to assess the applicability of suggested thresholds for clustering of potentially related isolates. Therefore, meat-associated isolates originating from the same samples or processing plants as well as fish-associated isolates were analyzed as distinct sets. In silico serogrouping, multilocus sequence typing (MLST), core genome MLST (cgMLST), and pangenome analysis were combined with screenings for prophages and genetic traits. Isolates of the same subtypes (cgMLST types (CTs) or MLST sequence types (STs)) were additionally compared by SNP calling. This revealed the occurrence of more than one CT within all three investigated plants and within two samples. Analysis of the fish set resulted in predominant assignment of isolates from pangasius catfish and salmon to ST2 and ST121, respectively, potentially indicating persistence within the respective production chains. The approach not only allowed the detection of distinct subtypes but also the determination of differences between closely related isolates, which need to be considered when interpreting WGS data for surveillance.
Posted in food contamination, food handler, Food Hazard, Food Hygiene, Food Inspections, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Pathogen, food recall, Food Safety, Food Safety Alert, Food Safety Management, Food Testing, Listeria, Listeria monocytogenes, MLST, Research, WGS
Posted in Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Research, Salmonella, Salmonella in Chicken
Invasive listeriosis is a potentially fatal foodborne disease that according to this study may affect up to 32.9 % of the US population considered as increased risk and including people with underlying conditions and co-morbidities. Listeria monocytogenes has been scrutinized in research and surveillance programs worldwide in Ready-to-Eat (RTE) food commodities (RTE salads, deli meats, soft/semi-soft cheese, seafood) and frozen vegetables in the last 30 years with an estimated overall prevalence of 1.4–9.9 % worldwide (WD) and 0.5–3.8 % in the United States (US). Current L. monocytogenes control efforts have led to a prevalence reduction in the last 5 years of 4.9–62.9 % (WD) and 12.4–92.7 % (US). A quantitative risk assessment model was developed, estimating the probability of infection in the US susceptible population to be 10–10,000× higher than general population and the total number of estimated cases in the US was 1044 and 2089 cases by using the FAO/WHO and Pouillot dose-response models. Most cases were attributed to deli meats (>90 % of cases) followed by RTE salads (3.9–4.5 %), soft and semi-soft cheese and RTE seafood (0.5–1.0 %) and frozen vegetables (0.2–0.3 %). Cases attributed to the increased risk population corresponded to 96.6–98.0 % of the total cases with the highly susceptible population responsible for 46.9–80.1 % of the cases. Removing product lots with a concentration higher than 1 CFU/g reduced the prevalence of contamination by 15.7–88.3 % and number of cases by 55.9–100 %. Introducing lot-by-lot testing and defining allowable quantitative regulatory limits for low-risk RTE commodities may reduce the public health impact of L. monocytogenes and improve the availability of enumeration data.
Posted in food contamination, food handler, Food Hazard, Food Hygiene, Food Inspections, Food Microbiology, Food Microbiology Research, Food Microbiology Testing, Listeria, Listeria monocytogenes, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Research
Executive summary
Shiga toxin-producing Escherichia coli (STEC) are estimated to cause more than 1.2 million illnesses and 128 deaths globally each year. The previous work of FAO and WHO identified beef and other types of meats, dairy products and produce as significant risk factors for STEC infection. As such, at its 42nd Session, the Codex Alimentarius Commission (CAC) endorsed the Codex Committee on Food Hygiene’s (CCFH) recommendation for the development of guidelines for the control of STEC in beef, raw milk and cheese produced from raw milk, leafy greens and sprouts.
To facilitate this work, the CCFH requested that FAO and WHO Joint Expert Meeting on Microbiological Risk Assessment (JEMRA) provide scientific advice on the effectiveness and utility of control measures against STEC during primary production, processing and post-processing of raw meat, raw milk and raw milk cheeses.
During the meeting, the expert committee reviewed interventions for the control of STEC in cattle, raw beef and raw milk and raw milk cheese manufactured from cows’ milk, and also evaluated available evidence for other small ruminants (goat, sheep), swine and other animals (reindeer, yak, camelids, bison, buffalo and swine). The expert committee was tasked with scoring the degree of support for the effectiveness of interventions for the specific control of STEC as high, medium or low based on the evidence available within the scientific literature.
In meat production and processing systems, many approaches to support control of STEC are based on good agricultural practices (GAP) and/or good hygiene practice (GHP) that aim to generally reduce the spread of pathogens and are not specifically focused on STEC. On-farm, these include managing the hygienic conditions of housing, bedding and drinking water hygiene, appropriate animal density and biosecurity measures, effective sanitation of facilities and proper disposal of manure.
On-farm, several dietary and herd management strategies with varying levels of impact on STEC populations in beef and dairy animals have been explored. Evidence to support cattle demography
(Section 2.1.3), animal density
(Section 2.2.2), biosecurity
(Section 2.2.1), and environmental hygiene
(Section 2.2.3) were rated as having a medium or medium to high degree of support with regards to their ability to impact STEC. Interventions including feeding of forage versus concentrate rations, specific grain types
(Section 2.3.3), and the inclusion of citrus products and essential oils in feed
(Section 4.2.5) were supported at low to medium or medium degree of support, yet probiotics may be useful with administered to cattle, goats and sheep through feed
(Sections 2.3.4.1 and 6.1.1). Some vaccines have been shown to reduce faecal excretion of STEC O157:H7
(Section 2.4.1), but their efficacy is variable depending on the vaccine and the number of doses administered.
Long distance transport and the stress of interim unloading/loading have been shown to increase faecal excretion of STEC that can lead to cross-contamination between animals
(Section 2.6). Transport distances should be minimized in accordance with best practices for animal welfare, and the evidence related specifically to the control of STEC was supported at a low degree. A summary of primary production control
measures for STEC in cattle and their degree of support rating (high, medium, low), based on scientific evidence, is available in Annex 1.
Avoiding contamination of the carcass through contact with hides, gut contents or faeces during slaughter is an accepted management practice during meat processing, but evidence supporting the effectiveness and reliability of these
measures for the control of STEC was limited. Processing measures where evidence supported a high or medium to high rating for efficacy in STEC reduction included steam vacuuming of visible faecal contamination on carcasses (Section 3.3.4.3),
and the use of a hot potable water carcass wash, steam pasteurization followed by 24 h air chilling and combinations of these
(Section 3.4). The use of knife trimming to remove carcass tissue contaminated with faecal material is common and is supported by a medium confidence level in the evidence
(Section 3.3.4.2). Despite the commercial use of pre-chill carcass decontamination treatments using organic acids and other chemical agents, the confidence in the evidence was low in cattle and other small ruminants due to high variability in results
(Section 3.4.3). A summary of processing control measures for STEC in beef and their degree of support (high, medium, low), based on scientific evidence, is available in Annex 2.
The efficacy of available control measures for reducing or eliminating STEC on primal cuts, trim, cheek meats, and ground beef was widely varied. Yet, the use chemical antimicrobial dips
(Section 4.2) for primals and trims were supported at a low to medium level of confidence, and high-pressure processing (HPP)
(Section 4.1.6), gamma irradiation and electron beam sterilization (eBeam)
(Section 4.1.7) produced significant reductions of STEC in ground beef and in retail packs. A summary of post-processing control measures, and combinations of these, for STEC in beef and their degree of support (high, medium, low), based on scientific
evidence, is available in Annex 3.
Pork products and meat from wild game have occasionally been confirmed as vehicles of STEC transmission, but there are no interventions or practices during the processing of these animals that are specific for STEC. Meat from these species could be treated post-harvest in a similar fashion as beef to reduce STEC, but reports of the efficacy of these interventions are not available.
Contamination of milk with pathogens, including STEC, mainly occurs during milking or via milking equipment, milking personnel, and from the farm environment. Thus, factors affecting the carriage of STEC in live animals and those practices surrounding milking hygiene can reduce, but not assure the absence of contamination of raw milk.
The efficacy of the interventions against STEC during the production of raw milk and raw milk cheeses varied greatly depending on the animal origin of the raw milk, manufacturing practices, the scale of production, and the microbial load. Temperature control and hygiene during milking, storage and transportation can significantly affect the microbiological safety of raw milk prior to processing, packaging and sale of milk intended for drinking or for manufacturing of raw milk cheeses. Although these interventions can mitigate the growth of E. coli and other indicator organisms, the degree of support in the evidence for these interventions and the control of STEC ranged from low to medium
(Section 2.5). Apart from pasteurization, which is very effective, several technologies have been evaluated to mitigate the presence of STEC in raw milk. Bacteriophages specific to E. coli and STEC have shown some reductions in STEC during refrigeration storage of raw milk
(Section 5.1.5). The effect of adding bacteriophage to control E. coli during milk fermentation in the making of cheeses has also been examined with varying results depending on the STEC serovar. The degree of support in the evidence of bacteriophage to specifically control for STEC was evaluated as low
(Section 5.2.3). Gamma or eBeam irradiation are very effective at reducing bacterial levels in milk and on cheese surfaces, yet off-flavors are often reported. The degree of support for the evidence was rated as medium
(Section 5.3.2). A summary of processing and post-processing control measures for STEC in raw milk and raw milk cheese and their degree of support rating (high, medium, low), based on scientific evidence, is available in Annex 4.
The implementation of monitoring plans at the farm level to measure the impact of STEC prevalence is considered impractical, although sampling and testing of beef and raw milk products are a means to verify that food safety program are successful. Because STEC are often present only at low levels in foods, culture enrichment of food samples is a critical step in detecting STEC in meat, dairy and other foods. Since STEC testing is complex, the quantitative detection of non-type specific (NTS) E. coli has been proposed as an alternative hygienic indicator during processing and post-processing stages, although it is not an absolute estimate of STEC levels.
The use of molecular techniques, such as PCR, that target STEC virulence genes are highly sensitive and specific for STEC detection but presumptive results must be confirmed by traditional culture-based methods or by immunomagnetic
separation (IMS). Methods are needed that enable the efficient and specific isolation of STEC O157:H7 and non-O157 STEC.
The expert committee also discussed some of the limitations and gaps regarding the available data. In-plant scientific evaluations of interventions and treatments to control STEC throughout raw beef, raw milk and raw milk cheese production are
frequently prohibited due to health risks associated with the potential introduction of pathogens into the food supply and the cost associated with testing large number of samples required for detecting STEC in food matrices. Consequently, surrogate
bacteria, such as NTS E. coli, are used as substitutes and the results extrapolated, meaning that evidence of intervention effects specifically for STEC may not be available currently or in the future. Therefore, there is doubt and uncertainty as to
whether the detection and reduction levels observed in surrogate studies are truly representative of STEC or of commercial production and processing.
Many studies focused on the impact of an individual control measure at a specific stage in the food chain, rather than in the context a total food chain or of the safety of the food available to the consumer. Many food businesses have implemented
multiple control measures concurrently or sequentially on farms and in processing facilities, but the overall efficacy of multiple “hurdles” in the total chain remains difficult to quantify
It was recognized that with advances in analytical methods, including increasing use of molecular tools, the evaluation of evidence concerning some STEC control measures and interventions may need to be revised in the future.
Posted in Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Research, STEC, STEC E.coli
Posted in Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Listeria, Listeria monocytogenes, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Research