Brand : Ambrosio
Name : Natural sweet almonds 36/38
Reason for reporting : Recall due to chemical risk
Publication date : 20 February 2023

Brand : Ambrosio
Name : Natural sweet almonds 36/38
Reason for reporting : Recall due to chemical risk
Publication date : 20 February 2023

Posted in Aflatoxin, Aspergillus Toxin, food contamination, food handler, Food Hazard, Food Hygiene, Food Inspections, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Poisoning, food recall, Food Safety, Food Safety Alert, Food Safety Management, Food Temperature Abuse, Food Testing, Food Toxin, Mold Toxin, Mould Toxin, Mycotoxin
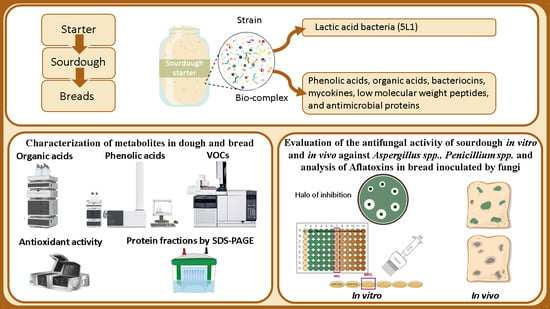
Posted in antifungal, Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, fungi, LAB, lactic acid bacteria, Lactobacillus, Lactobacillus plantarum, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk
Abstract
Ocimum gratissimum essential oil (EOGT) has been evaluated for its antibacterial efficacy, and its combinational therapy with antibiotics may enhance the therapeutic efficiency against infection-causing bacteria. Herein, we evaluated the chemical composition of EOGT and its antibiotic efficacy against Staphylococcus aureus and Pseudomonas aeruginosa. GC-MS and GC-FID analyzed EOGT. The antibiotic efficacy was determined by the agar diffusion method, microdilution, minimum inhibitory concentration (MIC), and fractional inhibitory concentration index (FICI). Eugenol (74.2%) was the main component of OGT. Using the agar diffusion method, the action of rifampicin, ciprofloxacin, and tetracycline was evaluated against S. aureus, while the action of cefepime and ciprofloxacin was evaluated against P. aeruginosa. FICI showed a reduced MIC of ciprofloxacin and tetracycline associated with EOGT. In the presence of EOGT, MIC of ciprofloxacin reduced from 0.6 to 0.0006 μg/mL and of tetracycline decreased from 0.028 to 0.0018 μg/mL against S. aureus and from 4 to 0.12 μg/mL against P. aeruginosa. EOGT enhanced the antibacterial efficacy of the antibiotics suggesting a synergistic effect, thereby enhancing the efficacy in treating infection against S. aureus and P. aeruginosa.
Posted in Antimicrobials, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Pseudomonas aeruginosa, Staphylococcus aureus
Posted in Aspergillus Toxin, Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Toxin, HACCP, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Mold Toxin, Mould Toxin, Mycotoxin
Posted in Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Food Pathogen, Listeria, Listeria monocytogenes, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Pathogen, pathogenic
Posted in Bacteriophage, Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Phage, Salmonella
Posted in Antibiotic Resistance, Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, Listeria monocytogenes, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk
Vaccine programs in the turkey industry are a primary pre-harvest Salmonella mitigation strategy, and development of effective programs rely heavily on effective Salmonella serovar surveillance. Noteworthy hurdles to surveillance and vaccine program development includes the fact that traditional isolation identifies only the most abundant serovars in a population, while underlying serovars remain unknown. Further, there is a lack of understanding where in the supply chain samples should be taken to inform serovars present in the system.
USPOULTRY and the USPOULTRY Foundation recently announced the completion of a funded research project at the University of Georgia in which researchers evaluated methods to improve Salmonella surveillance in turkeys. The research was made possible in part by an endowing Foundation gift from Cargill and proceeds from the International Poultry Expo, part of the International Production & Processing Expo (IPPE). The research is part of the Association’s comprehensive research program encompassing all phases of poultry and egg production and processing.
Posted in Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Salmonella
Posted in Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Salmonella, Vibrio
Posted in Antibacterial, Antimicrobials, Decontamination Microbial, Food Micro Blog, Food Microbiology, Food Microbiology Blog, Food Microbiology Research, Food Microbiology Testing, microbial contamination, Microbial growth, Microbiological Risk Assessment, Microbiology, Microbiology Investigations, Microbiology Risk, Salmonella, Staphylococcus aureus