MDPI
Recent shifts in food production, processing and distribution, linked to the globalization of the food trade and the need to meet new consumers habits, are continuously challenging global food systems. Every effort is being made to ensure healthy and safe, food that is crucial to guarantee public health and wellbeing.
Despite the advancements in food safety management, foodborne diseases (FBD) still remain an important problem worldwide, with a significant negative impact on human health and countries’ economies and development. It has been determined that food unsafe for consumption causes 600 million cases of FBD every year, and 25% of all foods produced globally are lost due to microbial spoilage. Serious outbreaks have occurred, involving both developing and industrialized countries, showing how food safety is a transnational challenge and that a strong joint commitment between food safety authorities is needed.
Despite this awareness, the full extent of the impact of food contamination is still unknown. Foodborne contaminants are numerous, including viruses and bacteria, parasites, chemicals, toxins and allergens that cause a wide range of conditions. Globally, FBD caused by bacteria are more common than those caused by viruses and parasites. [
1]. Moreover, between one-third and one-half of all human infectious diseases have a zoonotic origin. Among bacteria,
Campylobacter, followed by
Salmonella, are the major etiological agents of FBD, while, among viruses,
norovirus is the foremost enteric pathogen of foodborne disease worldwide [
2].
The most common foodborne parasites instead are protozoa such as
Cryptosporidium spp.,
Giardia intestinalis and
Toxoplasma gondii; roundworms such as
Trichinella spp. and
Anisakis spp.; and tapeworms such as
Diphyllobothrium spp. and
Taenia spp. [
3]. In particular, Anisakiasis is an emerging zoonosis caused by the fish parasitic nematode
Anisakis. Humans are accidental hosts that become infected by eating raw or undercooked fish that contain viable
Anisakis spp. larvae.
The major determinants for the incidence of FBD are unsafe raw food, abused temperature, inadequate storage, improper handling, undercooking and cross contamination [
4]. Food from animal sources, fresh produce and ready-to-eat (RTE) foods are the most at risk. In particular, RTE foods are an emerging issue concerning food safety. Furthermore, they have been demonstrated to contain antimicrobial-resistant strains. Since these products are consumed without any further treatment, they could serve as a vector for the spread of antibiotic-resistant microorganisms, posing a significant threat to public health [
5].
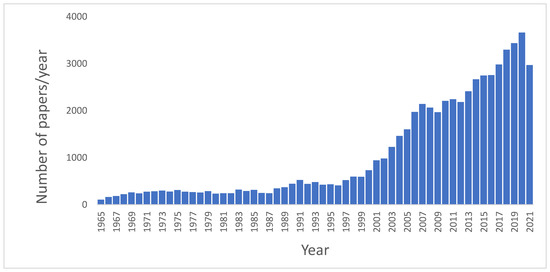
The importance of these topics is documented by the increasing number of papers published related to Food Safety. In a basic search using PubMed database, from 1945 to 2021, selecting as the search topic “Microbiological food safety”, a total of 54,210 results were obtained.
Although the first articles concerning microbiological food safety date from 1946, and since then a dozen articles appear in the following years, it is not until 1965 that a significant number of articles are published every year. shows the evolution of the number of papers per year (from 1965 to 2021) published regarding Microbiological food safety. As can be seen in this figure, approximately 80% of these papers have been published in the last 20 years, and the number continues to rise, revealing an increasing and ever-present interest towards the topic addressed in this Special Issue.