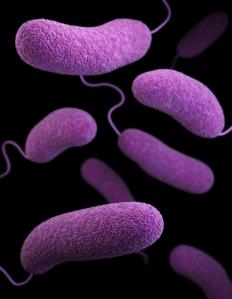
The U.S. Food and Drug Administration continues to help ensure the safety of powdered infant formula in the wake of reports of Cronobacter sakazakii infections in four infants that may have contributed to death in two patients.
Those reports led to an FDA investigation of Abbott Nutrition in Sturgis, Michigan. Insanitary conditions found at the facility led to the February 2022 voluntary recall of specific lots of powdered infant formula manufactured at the facility and brought attention to a type of bacteria that many parents may not have heard about before and scientists continue to work to better understand.
While Cronobacter contamination and infections are rare, it is still important to know the risks and what you can do to minimize them.
What Is Cronobacter?
Cronobacter is a germ (bacteria) that is naturally found in the environment. Cronobacter can exist on almost any surface and is especially good at surviving in dry foods, like powdered infant formula, powdered milk, herbal teas, and starches.
Cronobacter is harmless for most people and infections are rare. When they do occur, they can be life threatening in infants, particularly those who are younger than two months old, premature, immunocompromised, or of low birthweight.
How Does Powdered Infant Formula Become Contaminated with Cronobacter?
Parents may not know that while powdered infant formula manufacturers are required to produce formula that is safe from harmful bacteria like Cronobacter, powdered formulas — unlike liquid formulas — do not receive certain processing treatments that would render them sterile.
Cronobacter has the potential to come into factories, homes, and hospitals on the soles of shoes or on hands and can then live on surfaces like counters or bottles.
At the factory, Cronobacter could get into formula powder if:
- Contaminated ingredients are used to make the formula.
- The formula powder touches a contaminated surface.
In the home, infants can get infected by Cronobacter through multiple pathways including:
- Inadequately sanitized breast-pump accessories, bottles, and other utensils used for feeding.
- Inadequate handwashing prior to infant formula preparation.
- Formula lids or scoops placed on contaminated surfaces, like countertops or sinks, that then later touch the formula.
- Contaminated water used to mix formula.
Why Is it Difficult to Find the Source of Cronobacter Contamination?
Cronobacter sakazakii and its cousins in the broader Cronobacter species category are part of a large family of bacteria that are common in the environment — like in our yards, kitchens, and living rooms — so identifying the specific source of an illness or outbreak can be challenging.
Importantly, only one state in the U.S., Minnesota, currently requires that cases of Cronobacter sakazakii be reported to state public health authorities. As a result, many cases are unreported, and scientists have limited information about the genetic makeup of the bacteria found in cases that are reported. To provide some perspective, the National Center for Biotechnology Informatics (NCBI) database that tracks these pathogens has received roughly 480,000 submissions of genomic information for strains of Salmonella enterica compared to only 1086 submissions for strains of Cronobacter.
What Can You Do to Minimize the Risk of Cronobacter Contamination?
Safely Preparing and Storing Powdered Infant Formula
In most cases, it is safe to mix powdered infant formula following manufacturer’s instructions. If your baby is less than 2 months old, was born prematurely, or has a weakened immune system the Centers for Disease Control and Prevention (CDC) recommends using ready-to-feed infant formula. Liquid infant formula is made to be sterile (without germs) and is the safest option for infants not receiving breast milk. However, you can also take extra steps to prepare powdered formula for these infant groups by using hot water (at least 158°F/70°C) to help protect against Cronobacter and then cooling the formula before feeding your infant.
However, certain metabolic and specialty products include statements on their packaging warning consumers against heating because heating the particular product above 100°F could result in a loss of vitamins and nutrients. Therefore, caregivers should be especially mindful of the manufacturer’s instructions included on the packaging for specialty metabolic formulas. If the instructions prevent a consumer from heating their formula to at least 158°F, then caregivers should instead bring the water used to mix the formula to body temperature and follow the instructions in this printable infographic to prepare and store powdered infant formula safely.
Parents who suspect their infant became ill due to infant formula may call the FDA’s consumer hotline at 1-800-FDA-1088.
Other Tips to Minimize the Risk of Cronobacter Contamination
Infants could also be exposed to Cronobacter from unclean breast pumps or unclean bottles. To help keep your baby safe, you can help prevent Cronobacter from growing on these items. Keep your baby’s food, whether breast milk or formula, safe by carefully cleaning, sanitizing, and storing bottles and breast pump parts.
It is also important to wash your hands with soap and water, especially before preparing bottles and feeding. The CDC says that alcohol-based hand sanitizer with at least 60% alcohol can also be used.
What Are the Symptoms of Cronobacter infection?
Symptoms of infection in infants may start with fever, poor feeding, excessive crying, and/or very low energy. Some infants may also have seizures. Babies with these symptoms should be taken to a medical provider as soon as possible.