FDA
Company Announcement
FOR IMMEDIATE RELEASE –Monterey, CA–January 7, 2022–Dole Fresh Vegetables, Inc. is voluntarily recalling from the market all Dole-branded and private label packaged salads described below processed at its Springfield, OH and Soledad, CA production facilities containing iceberg lettuce, due to a possible health risk from Listeria monocytogenes.
Listeria monocytogenes is an organism that can cause serious and sometimes fatal infections in young children, frail or elderly people, and others with weakened immune systems. Although healthy individuals may suffer only short-term symptoms such as high fever, severe headache, stiffness, nausea, abdominal pain and diarrhea, Listeria infection can cause miscarriages and stillbirths among pregnant women.
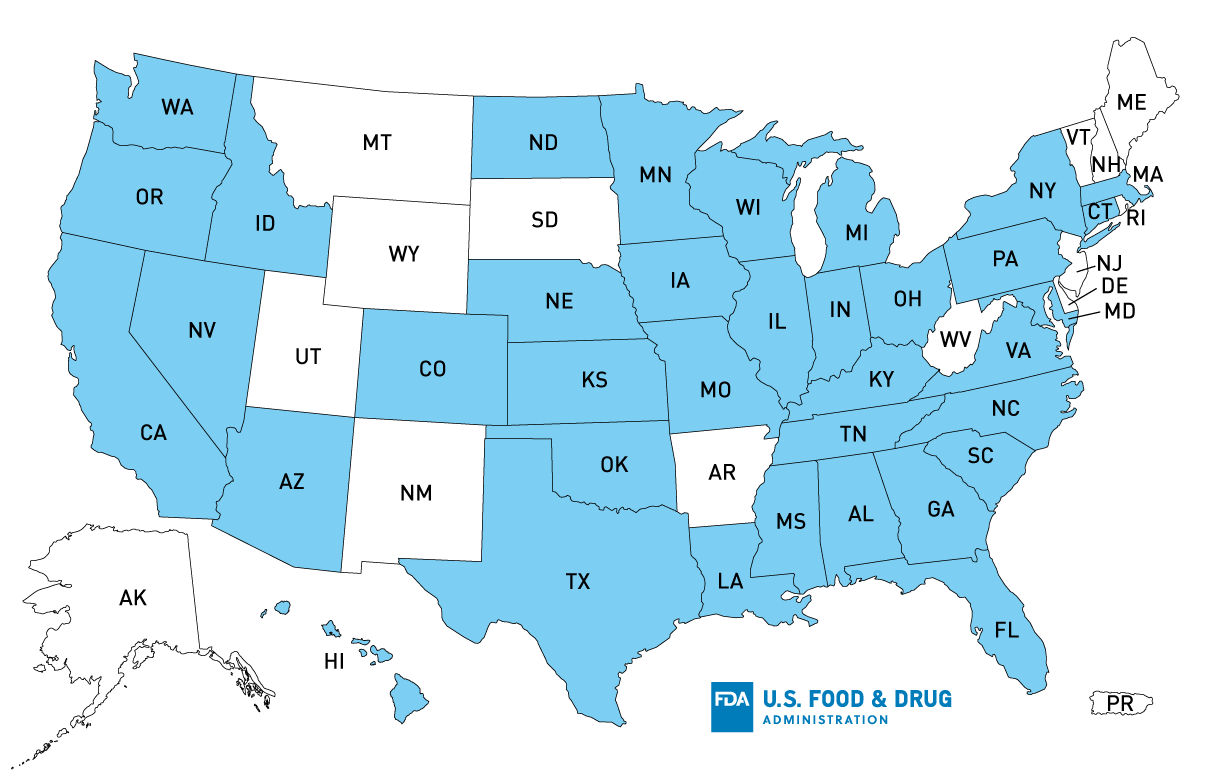
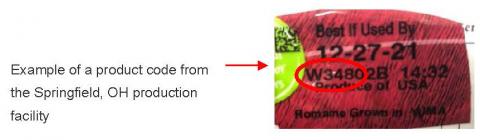
Products subject to the voluntary recall from Springfield, OH are identified by a product lot code beginning with the letter “W” and a “Best if Used By” date between December 22, 2021 and January 9, 2022. Products subject to the voluntary recall from Soledad, CA are identified by a product lot code beginning with the letter “B” and a “Best if Used By” date between December 23, 2021 and January 8, 2022. The product lot codes are located in the upper-right-hand corner of the package (see example below). Consumers who still have any of these products in their refrigerators are urged not to consume the product and to discard it immediately. No illnesses have been reported with the products being recalled to date.
This voluntary recall notification is being issued after harvest equipment used in the harvesting of the raw iceberg lettuce material used in these finished products was tested by Dole and found to contain Listeria monocytogenes.
Listing of products subject to each of these recalls is set forth below.
Recalled salad items from the Springfield facility were distributed in the states of AL, CO, CT, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MO, NC, NE, NY, OH, PA, SC, TN, VA and WI. Additionally, these salads were distributed in the following Canadian provinces: New Brunswick, Ontario, and Quebec. Recalled salad items from the Soledad facility were distributed in the states of AL, AZ, CA, CO, FL, GA, HI, IA, ID, IL, IN, KS, LA, MI, MN, MO, MS, MD, ND, NE, NV, NY, OK, OR, PA, TX, UT, VA, WA and WI. Additionally, these salads were distributed in the following Canadian provinces: Alberta, British Columbia, and Saskatchewan.
No other Dole products, including fresh fruit and field-packed fresh vegetables, are part of these voluntary recalls and are safe to consume.
Dole retailers have been advised to check store shelves and warehouse inventories to confirm that no recalled product is available for purchase by consumers.
Retailer and consumer questions about the voluntary recalls should be directed to the Dole Consumer Response Center at 800-356-3111, Monday-Friday, 8:00am to 3:00pm Pacific Time.
Dole Fresh Vegetables is coordinating closely with regulatory officials.
Product Listing on the following pages
LISTING OF PRODUCTS SUBJECT TO SOLEDAD RECALL
| Product Description |
UPC Code |
| Dole 10oz Very Veggie |
0-71430-01008-2 |
| Dole 11oz Greener Selection |
0-71430-00965-9 |
| Dole 12oz American |
0-71430-00933-8 |
| Dole 12oz Garden Salad / Salade Du Jardin |
0-71430-01135-5 |
| Dole 13.25oz Country Ranch Kit |
0-71430-01730-2 |
| Dole 13oz Southwest Kit |
0-71430-01701-2 |
| Dole 16oz Value Size Shredded Lettuce |
0-71430-84616-2 |
| Dole 24oz Value Size Garden Salad |
0-71430-01136-2 |
| Dole 3lb Garden Salad |
0-71430-01138-6 |
| Dole 5.8 oz Chicken Club BLT Salad Bowl |
0-71430-00123-3 |
| Dole 6.05 oz Avocado Ranch Salad with Chicken Bowl |
0-71430-00125-7 |
| Dole 6.25 oz Backyard BBQ Salad with Chicken Bowl |
0-71430-00124-0 |
| Dole 6.25 oz Santa Fe Style Salad Bowl |
0-71430-00118-9 |
| Dole 7.25 oz Turkey & Bacon Country Cobb Salad Bowl |
0-71430-00120-2 |
| Dole 7.65oz Café Chef Salad Bowl |
0-71430-00115-8 |
| Dole 8oz Shredded Lettuce |
0-71430-01065-5 |
| Dole 9.1oz Peppercorn Ranch Chopped Kit |
0-71430-00093-9 |
| Dole 9.8oz Chopped Teriyaki Pineapple Kit |
0-71430-00100-4 |
| HEB 12oz American Salad |
0-41220-35500-6 |
| HEB 8oz Shredded Lettuce |
0-41220-35521-1 |
| HEB 9.5oz Premium Ranch Kit |
0-41220791569-0 |
| Marketside 7.45 oz Premium Avocado Ranch Bowl |
6-81131-37735-5 |
| Marketside 11.75 oz Multi-Serve Southwestern Style Salad Bowl |
6-81131-42361-8 |
| Marketside 12oz Classic Salad |
6-81131-32894-4 |
| Marketside 12oz Crisp Greens |
6-81131-35503-2 |
| Marketside 1lb Shredded Lettuce |
6-81131-53209-9 |
| Marketside 24oz Classic Salad |
6-81131-32895-1 |
| Marketside 6.35 oz Santa Fe Style Bowl |
6-81131-42363-2 |
| Marketside 6.81 oz BLT Salad Bowl |
6-81131-37736-2 |
| Marketside 7.25 oz Chef Salad Bowl |
6-81131-35506-3 |
| Marketside 7.25 oz Cobb Salad Bowl |
6-81131-35507-0 |
| Marketside 8oz Shredded Lettuce |
6-81131-32896-8 |
| Presidents Choice 12.25oz Ranch Kit / Le Choix du President Ranch Kit de Salade |
0-60383-00493-4 |
| President’s Choice 12oz Garden Salad / Le Choix du President Salade Jardiniere |
0-60383-22268-0 |
| President’s Choice 13oz Sesame Ginger Kit / Le Choix du President Sesame et Gingembre |
0-60383-02320-1 |
| President’s Choice 13oz Southwest Salad Kit / Le Choix du President Sud-Ouest |
0-60383-02319-5 |
LISTING OF PRODUCTS SUBJECT TO SPRINGFIELD RECALL
| Product Description |
UPC Code |
| Dole 10 oz Very Veggie / Ranache de Legumes |
0-71430-01008-2 |
| Dole 11 oz Greener Selection / Selection de Verdure |
0-71430-00965-9 |
| Dole 12 oz American / Melange Americain |
0-71430-00933-8 |
| Dole 12 oz Garden Salad / Salade du Jardin |
0-71430-01135-5 |
| Dole 13 oz Asian Island Crunch / Croustillante Des Iles Asiatiques |
0-71430-01721-0 |
| Dole 13 oz Southwest Salad Kit |
0-71430-01701-2 |
| Dole 13 oz Southwest Salad Kit / Salade du Sud-ouest |
0-71430-01711-1 |
| Dole 13.25 Country Ranch Kit |
0-71430-01730-2 |
| Dole 16 oz Shredded Lettuce |
0-71430-84616-2 |
| Dole 2 lb Garden Salad |
0-71430-01137-9 |
| Dole 24 oz Value Size Garden Salad |
0-71430-01136-2 |
| Dole 8 oz Shredded Lettuce |
0-71430-01065-5 |
| Dole 9.1 oz Peppercorn Ranch / Ranch et Poivre |
0-71430-00098-4 |
| Dole 9.1 oz Peppercorn Ranch Chopped Kit |
0-71430-00093-9 |
| Dole 9.8 oz Chopped Teriyaki Pineapple Kit |
0-71430-00100-4 |
| Dole 9.8 oz Chopped Teriyaki Pineapple Kit / Teriyaki A L’ananas |
0-71430-00182-0 |
| Kroger 12 oz American Blend |
0-11110-91041-7 |
| Kroger 12 oz Classic Garden |
0-11110-91036-3 |
| Kroger 12 oz Veggie Blend |
0-11110-91048-6 |
| Kroger 24 oz Classic Garden |
0-11110-91037-0 |
| Kroger 8oz. Shredded Iceberg Lettuce |
0-11110-91613-6 |
| Little Salad Bar 10 oz Caesar Salad Kit |
4099100 087000 |
| Little Salad Bar 10.65 oz. Chopped Caesar Kit |
4099100263923 |
| Little Salad Bar 12 oz Garden Salad |
4099100082982 |
| Little Salad Bar 8 oz Shredded Lettuce |
4099100087963 |
| Little Salad Bar 9 oz Italian Salad |
4099100083194 |
| Marketside 12 oz Classic Iceberg Salad |
6-81131-32894-4 |
| Marketside 12 oz Crisp Greens |
6-81131-35503-2 |
| Marketside 16 oz Shredded Lettuce |
6-81131-53209-9 |
| Marketside 24 oz Classic Iceberg Salad |
6-81131-32895-1 |
| Marketside 8 oz Shredded Lettuce |
6-81131-32896-8 |
| Presidents Choice 12 oz Garden Salad / Le Choix du President Salade Jardiniere |
0-60383-22268-0 |
| Presidents Choice 12 oz Mixed Greens / Le Choix du President Melange de Legumes- Feuilles |
0-60383-00188-9 |
| Presidents Choice 12.25 oz Ranch Kit / Le Choix du President Ranch Kit de Salade |
0-60383-00493-4 |
| Presidents Choice 13 oz. Sesame Ginger Kit / Le Choix du President Sesame et Gingembre |
0-60383-02320-1 |
| Presidents Choice 13 oz. Southwest Salad Kit / Le Choix du President Sud-Ouest |
0-60383-02319-5 |
Company Contact Information
- Consumers:
- Dole Consumer Response Center
- 800-356-3111
- Media:
- Bil Goldfield
- 818-874-4647