Abstract
Practical Application
Listeria monocytogenes constitutes a major problem for the food industry due to its widespread availability in nature and its role as an environmental contaminant in food processing plants.
Listeria monocytogenes constitutes a major problem for the food industry due to its widespread availability in nature and its role as an environmental contaminant in food processing plants.
The study evaluated the occurrence of Listeria spp. in three dairy plants located in Southern Brazil, as well as characterized the L. monocytogenes isolates according to serotypes, virulence genes and PFGE. The presence of Listeria species indicated postprocessing contamination in the dairy plants and threat to public health. Furthermore, the isolates showed a low genetic diversity, since only two pulsotypes were identified, highlighting the strain’s persistence in the processing environment and/or the presence of clones in this region.
Ottawa, December 4, 2019 – Usine Amsellem Inc. is recalling Amsellem brand Solo Chorizo – Dried Beef Sausage from the marketplace due to possible Salmonella contamination. Consumers should not consume the recalled product described below.
| Brand | Product | Size | UPC | Codes |
|---|---|---|---|---|
| Amsellem | Solo Chorizo – Dried Beef Sausage | 28 g | 6 28055 38930 9 | 91002DC-707 BB 2020 AVR 08 |
There have been no reported illnesses associated with the consumption of this product.
The bacterial pathogen Shigella, often spread through contaminated food or water, is a leading cause of mortality in both children and older adults in the developing world. Although scientists have been studying Shigella for decades, no effective vaccine has been developed, and the pathogen has acquired resistance to many antibiotics. The recent discovery of an early adherence step in the infection cycle by researchers at Massachusetts General Hospital (MGH) could provide a new therapeutic target or even a new method for vaccine development.
As it moves through the digestive system, Shigella traverses the small intestine and subsequently infects the large intestine, causing cramping, diarrhea and dehydration in the disease called shigellosis. “We wanted to determine how Shigella makes its first contact with epithelial cells in the early stages of disease development,” says Dr. Christina Faherty, senior author on the study published in mSphere. “Because of certain gene sequence annotations, and the way that Shigella appeared following growth in standard laboratory media, it was believed that Shigella strains do not produce fimbriae or other adherence factors.” Fimbriae are short hair-like fibers that bacterial cells use to adhere to individual epithelial cells to instigate infection.
Garlic extract not only has an antimicrobial activity but also has a stimulatory effect on the Lactobacillus spp. growth. On the other hand, some Lactobacillus strains can inhibit pathogenic bacteria. Then, the combination of Lactobacillus and garlic extract may be used to produce new bio‐preserved and functional meat products. The current study indicated the potential of Lactobacillus combined with garlic extract to control microbial and chemical changes in ground beef. The combination of Lactobacillus plantarum and garlic extract significantly (p < .05) reduced Listeria monocytogenes counts and lipid oxidation rates and improved the sensory scores in ground beef.
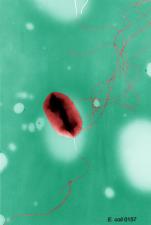
Image CDC
FDA, CDC, and state health authorities are investigating an outbreak of illnesses caused by E. coli O157:H7 in the United States. Epidemiologic, laboratory, and traceback evidence indicates that romaine lettuce from the Salinas, California growing region is a likely source of this outbreak. According to CDC, there have been 102 cases reported in 23 states. The latest date that one of these patients reports becoming ill was on November 18, 2019.
Consumers: Consumers should not eat romaine lettuce harvested from Salinas, California. Additionally, consumers should not eat products identified in the recall announced by the USDA on November 21, 2019.
Romaine lettuce may be voluntarily labeled with a harvest region. If this voluntary label indicates that the romaine lettuce was grown in “Salinas” (whether alone or with the name of another location) do not eat it. Throw it away or return it to the place of purchase. If romaine lettuce does not have information about harvest region or does not indicate that it has been grown indoors (i.e., hydroponically- and greenhouse-grown), throw it away or return it to the place of purchase. Consumers ordering salad containing romaine at a restaurant or at a salad bar should ask the staff whether the romaine came from Salinas. If it did, or they do not know, do not eat it.
At this time, romaine lettuce that was harvested outside of the Salinas region has not been implicated in this outbreak investigation. Hydroponically- and greenhouse-grown romaine, which is voluntarily labeled as “indoor grown,” from any region does not appear to be related to the current outbreak. There is no recommendation for consumers to avoid using romaine harvested from these other sources.
Restaurants and Retailers: Restaurants and retailers should not serve or sell romaine harvested from Salinas, California. If you do not know the source of your romaine lettuce, and if you cannot obtain that information from your supplier, you should not serve, nor sell it.
Suppliers and Distributors: Suppliers, distributors and others in the supply chain should not ship or sell romaine harvested in Salinas, California. If the source of the romaine lettuce is unknown, you should not ship, nor sell the product.
For Restaurants, Retailers, Suppliers and Distributors: Currently, the FDA does not have enough traceback information to identify the specific source of the contamination that would allow us to request a targeted recall from specific growers.
FDA requested that industry voluntarily withdraw romaine grown in Salinas from the market and is requesting that industry withhold distribution of Salinas romaine for the remainder of the growing season in Salinas. Without more specific traceback information, this was the most efficient way to ensure that contaminated romaine was off the market.
At this time, romaine lettuce that was harvested outside of the Salinas region has not been implicated in this outbreak investigation. Hydroponically- and greenhouse-grown romaine, which is voluntarily labeled as “indoor grown,” from any region does not appear to be related to the current outbreak. There is no recommendation for consumers or retailers to avoid using romaine harvested from these other sources.
According to the CDC, as of December 2, 2019, 102 people infected with the outbreak strain of E. coli O157:H7 have been reported from 23 states. The case patients report that illnesses started on dates ranging from September 24, 2019 to November 18, 2019. Illness onset dates reported to date are prior to the public warning issued on November 22.
Based on available traceback data, FDA requested that industry voluntarily withdraw romaine grown in Salinas from the market and is requesting that industry withhold distribution of Salinas romaine for the remainder of the growing season in Salinas. This was the most efficient way to ensure that contaminated romaine was off the market.
Products that were part of the U.S. Department of Agriculture’s Food Safety and Inspection Service (FSIS) announced recall related to this outbreak investigation had a “Best By” of Nov. 1 or earlier, more than one month ago, and should no longer be on the market.
FDA continues to actively investigate the cause of this outbreak. FDA, with the assistance of staff from the California Department of Public Health and the California Department of Food and Agriculture, immediately deployed investigators to three farms in the Salinas area that were identified based on the traceback investigation. On the farms, investigators sampled soil and animal droppings, compost, water, and other potential environmental sources. The samples and information collected during the farm investigations are currently being analyzed. Investigators are attempting to identify any factors that could have led to contamination. Additionally, state partners are testing romaine lettuce samples for E. coli that they have collected from stores and from case patients’ homes.
The FDA and state partners are conducting a traceback investigation to determine whether a common supplier or source of contamination can be identified. This investigation involves collecting and analyzing potentially hundreds of distribution records to trace the romaine that may have been available at points of exposure reported by ill people to their source. We are continuing to collect these records.
This remains an evolving and fluid situation. Information about our findings will be forthcoming as the investigation proceeds.
The Salinas region as defined by the United Fresh Produce Association and the Produce Marketing Association Romaine Taskforce ReportExternal Link Disclaimer includes: Santa Cruz, Santa Clara, San Benito, and Monterey counties in California.
Total Illnesses: 102
Hospitalizations: 58
Deaths: 0
Last illness onset: November 18, 2019
States with Cases: AZ (3), CA (4), CO (6), FL (1), IA (1), ID (3), IL (1), MD (4), MI (1), MN (3), MT (1), NE (1), NJ (7), NC (1), NM (2), OH (12), OR (1), PA (8), SD (1), TX (4), VA (4), WA (2), WI (31)
EP could be used for successful preservation of meat and meat products.
The Ministry for Primary Industries (MPI) has directed unregistered raw (unpasteurised) drinking milk suppliers across the country to stop selling their product until they comply with the legal requirements for sale.
MPI compliance staff yesterday executed search warrants at non-compliant raw drinking milk suppliers in Auckland, Hawkes Bay, Manawatu, Horowhenua, Nelson, and Southland following a year-long operation.
MPI’s manager of food compliance Melinda Sando says the purpose of the coordinated site visits was to gather evidence of the offending and to allow further investigation of non-compliant sales.
“We believe that the suppliers we visited today are operating outside of the regulatory framework. By not adhering to the rules for selling raw drinking milk, they are putting consumer health at risk.
“There have been multiple instances in the past of people getting sick after drinking raw milk from some of these suppliers. We can’t let this continue.
“Raw unpasteurised milk is a risky product as it hasn’t been heat-treated (pasteurised) to remove illness-causing bacteria including E. coli, listeria and Campylobacter.
“These types of bacteria most commonly cause severe diarrhoea and vomiting, but occasionally some have been linked with more serious complications that include miscarriage, paralysis, meningitis, and serious kidney problems in children. Raw milk may also be a source of tuberculosis (Tb).
The U.S. Food and Drug Administration (FDA), along with the Centers for Disease Control and Prevention (CDC), and state and local partners, are investigating a multistate outbreak of hepatitis A illnesses in Indiana, Michigan, Minnesota, Missouri, Nebraska, and Wisconsin potentially linked to fresh conventional (non-organic) blackberries from the grocery store, Fresh Thyme Farmers Market.
On December 3, 2019, CDC updated their case counts to 16 illnesses, with the most recent illness onset date on November 15, 2019.
Based on the epidemiological information collected in the investigation thus far, ill patients reported consuming fresh conventional blackberries from Fresh Thyme Farmers Market stores in six states: Indiana, Michigan, Minnesota, Missouri, Nebraska, and Wisconsin.
However, traceback information to date shows that these berries came from a distribution center that ships fresh berries to Fresh Thyme Farmers Market stores in 11 states: IA, IL, IN, KY, MI, MO, MN, NE, OH, PA, and WI. As this investigation continues, the FDA will work with our federal and state partners to obtain additional information during the traceback investigation and will update this advisory as more information becomes available.
Recommendation
The FDA is urging consumers to not eat any fresh conventional blackberries if purchased between September 9 and September 30, 2019, from Fresh Thyme Farmers Market stores in the 11 states mentioned above. People who purchased the fresh blackberries and then froze those berries for later consumption should not eat these berries. They should be thrown away.
If consumers purchased fresh conventional blackberries from Fresh Thyme Farmers Market stores in the 11 states listed above between September 9-30, ate those berries in the last two weeks, and have not been vaccinated for the hepatitis A virus (HAV), they should consult with their healthcare professional to determine whether post exposure prophylaxis (PEP) is indicated. PEP is recommended for unvaccinated people who have been exposed to HAV in the last two weeks. Those with evidence of previous hepatitis A vaccination or previous hepatitis A infection do not require PEP.
Contact your healthcare provider if you think you may have become ill from eating these blackberries, or if you believe that you have eaten these berries in the last two weeks.
Ottawa, December 3, 2019 – Loblaw Companies Ltd. is recalling President’s Choice brand Coleslaw from the marketplace due to possible Salmonella contamination. Consumers should not consume the recalled product described below.
| Brand | Product | Size | UPC | Codes |
|---|---|---|---|---|
| President’s Choice | Coleslaw | 397 g | 0 60383 22267 3 | Best Before 2019 DE 04 – B318005 and Best Before 2019 DE 04 – B318006 |
This recall was triggered by Canadian Food Inspection Agency (CFIA) test results. The CFIA is conducting a food safety investigation, which may lead to the recall of other products. If other high-risk products are recalled, the CFIA will notify the public through updated Food Recall Warnings.
The CFIA is verifying that industry is removing the recalled product from the marketplace.
There have been no reported illnesses associated with the consumption of this product.