Summary
- Company Announcement Date:
- FDA Publish Date:
- Product Type:
- Food & Beverages
Foodborne Illness - Reason for Announcement:
-
Potential contamination with shiga toxin-producing E. Coli
- Company Name:
- Cuisine Innovations Unlimited LLC
- Brand Name:
-
Earth Grown Vegan
- Product Description:
-
Earth Grown Vegan Traditional Falafel and Garlic & Herb Falafel
Company Announcement
Cuisine Innovations Unlimited, LLC of 180 Lehigh Avenue, Lakewood, NJ, is voluntarily recalling its Earth Grown Vegan Traditional Falafel and Garlic & Herb Falafel Distributed & Sold Exclusively by Aldi, Batavia, IL due to the possible presence of Shiga toxin-producing E. coli.
Shiga-toxin-producing E. coli is an organism that can cause foodborne illness in a person who eats a food item contaminated with it. Symptoms of infection may include stomach cramps, diarrhea, and vomiting. The illness primarily impacts elderly individuals, children, and people with weakened immune systems. Most healthy adults and children rarely become seriously ill.
The voluntarily recalled Earth Grown Vegan Traditional Falafel And Garlic & Herb Falafel Distributed & Sold Exclusively by Aldi, Batavia, IL were distributed in ALDI retail stores in the following states: Alabama, Arizona, Arkansas, California, Connecticut, Delaware, District of Columbia, Florida, Georgia, Illinois, Indiana, Iowa, Kansas, Kentucky, Louisiana, Maryland, Massachusetts, Michigan, Minnesota, Mississippi, Missouri, Nebraska, New Hampshire, New Jersey, New York, North Carolina, North Dakota, Ohio, Oklahoma, Pennsylvania, Rhode Island, South Carolina, South Dakota, Tennessee, Texas, Virginia, West Virginia, and Wisconsin.
The product comes in a bag in a box marked with any of the following Lot numbers: 1472, 1481, 1531, 1532, 1541, 1552, 1561, 1581, 1601, 1611, 1612, 1661, 1682, 1732, 1752, 1762, 1782, 1802, and 1812, and would have been shipped to stores after June 24, 2021. The lot number can be found in the “Best if used by” area on the outside flap of the box. There has been no further production of this product.
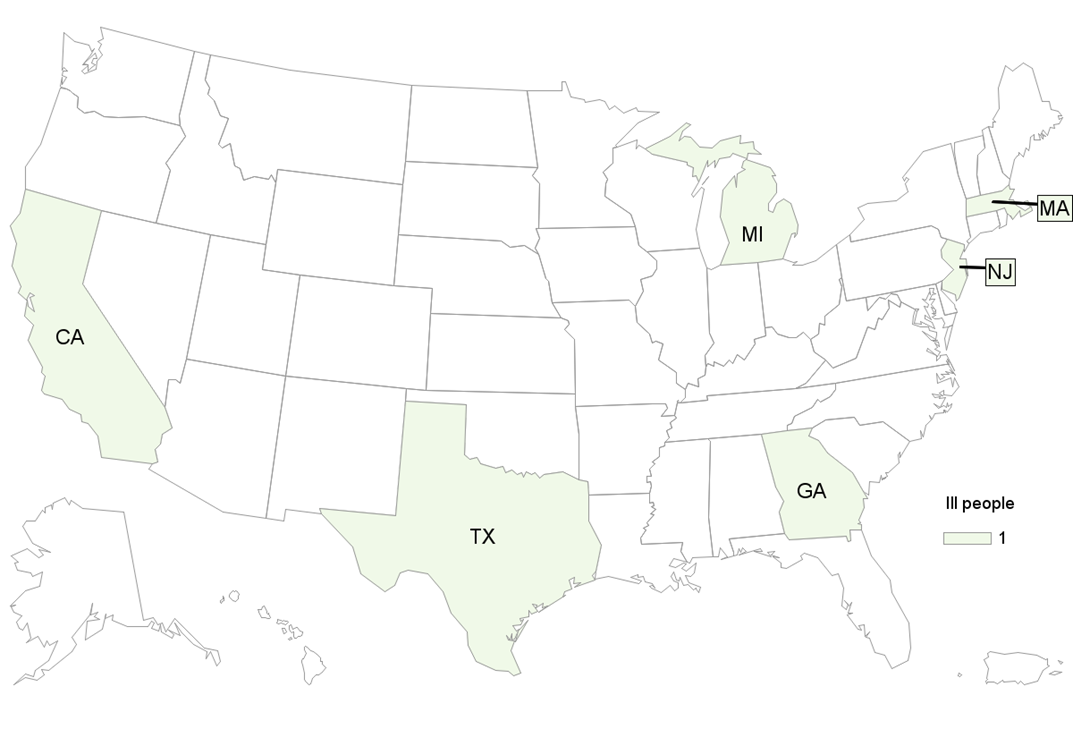
There have been 20 reported cases in six (6) states (FL, IA, KS, MI, OH and WI) with onset dates reported between July 24, 2022 and September 19, 2022 with 5 hospitalization and no deaths.
Consumers who have purchased Earth Grown Vegan Traditional Falafel and Garlic & Herb Falafel Distributed & Sold Exclusively by Aldi, Batavia, IL with any of the above lot numbers after June 24, 2021, are urged to return them to the place of purchase for a full refund. Consumers with questions may contact the company Monday through Friday, 8:00 a.m. to 4:00 p.m. Eastern Time at 1-201-439-1036, Ext. 26.
Company Contact Information
- Consumers:
- 1-201-439-1036, Ext. 26